1. Introduction
Obesity is quickly becoming the number one health issue confronting America today, and has also risen to epidemic proportions worldwide. Its spread has been associated with the adoption of a Western-style diet. However, I believe that the widespread consumption of food imports produced by U.S. companies plays a crucial role in the rise in obesity worldwide. Specifically, these "fast foods" typically include heavily processed derivatives of corn, soybeans, and grains, grown on highly efficient mega-farms. Furthermore, I will argue in this essay that one of the core underlying causes of obesity may be sulfur deficiency.
Sulfur is the eighth most common element by mass in the human body, behind oxygen, carbon, hydrogen, nitrogen, calcium, phosphorus, and potassium. The two sulfur-containing amino acids, methionine and cysteine, play essential physiological roles throughout the body. However, sulfur has been consistently overlooked in addressing the issues of nutritional deficiencies. In fact, the American Food and Drug Administration has not even assigned a minimum daily requirement (MDR) for sulfur. One consequence of sulfur's limbo nutritional status is that it is omitted from the long list of supplements that are commonly artificially added to popular foods like cereal.
Sulfur is found in a large number of foods, and, as a consequence, it is assumed that almost any diet would meet the minimum daily requirements. Excellent sources are eggs, onions, garlic, and leafy dark green vegetables like kale and broccoli. Meats, nuts, and seafood also contain sulfur. Methionine, an essential amino acid, in that we are unable to synthesize it ourselves, is found mainly in egg whites and fish. A diet high in grains like bread and cereal is likely to be deficient in sulfur. Increasingly, whole foods such as corn and soybeans are disassembled into component parts with chemical names, and then reassembled into heavily processed foods. Sulfur is lost along the way, and there is a lack of awareness that this matters.
Experts have recently become aware that sulfur depletion in the soil creates a serious deficiency for plants [Jez2008], brought about in part by improved efficiency in farming and in part, ironically, by successful attempts to clean up air pollution. Over the last two decades, the U.S. farming industry has steadily consolidated into highly technologized mega farms. The high yield per acre associated with these farms results in greater depletion of sulfur each year by the tall, densely planted crops. Plants require sulfur in the form of the sulfate radical (SO4-2). Bacteria in well aerated soil, similar to nitrogen fixing bacteria, can convert elemental sulfur into sulfate through an oxidation process. Coal contains a significant amount of sulfur, and factories that burn coal for energy release sulfur dioxide into the air. Over time, sun exposure converts the sulfur dioxide to sulfate, a significant contributor to acid rain. Acid rain is a serious pollutant, in that hydrogen sulfate, a potent acid, penetrates lakes, making them too acidic for lifeforms to thrive. The Clean Air Act, enacted by congress in 1980, has led to substantial decreases in the amount of acid rain released into the atmosphere. Factories have introduced highly effective scrubbing technologies to comply with the law, and, as a consequence, less sulfate makes its way back into the soil.
Modern farmers apply highly concentrated fertilizer to their soil, but this fertilizer is typically enriched in phosphates and often contains no sulfur. Excess phosphates interfere with sulfur absorption. In the past, organic matter and plant residues remained after the fruit and grain were harvested. Such accumulating organic matter used to be a major source of recyclable sulfur. However, many modern machinery-based methods remove a great deal more of the organic matter in addition to the edible portions of the plant. So the sulfur in the decaying organic matter is also lost.
It is estimated that humans obtain about 10% of their sulfur supply from drinking water. Remarkably, people who drink soft water have an increased risk to heart disease compared to people who drink hard water [Crawford1967]. Many possible reasons have been suggested for why this might be true ( Proposed theories for soft water/hard water differences in heart disease), and just about every trace metal has been considered as a possibility [Biorck1965]. However, I believe that the real reason may simply be that hard water is more likely to contain sulfur. The sulfate ion is the most useful form of sulfur for humans to ingest. Water softeners provide a convenient environment for sulfur-reducing bacteria, which convert sulfate (SO4-2) into sulfide (S-2), emitting hydrogen sulfide gas. Hydrogen sulfide gas is a poison that has been known to cause nausea, illness and, in extreme cases, death. When the bacteria are thriving, the gas will diffuse into the air and give off a foul odor. Obviously, it is rare that the concentration is sufficiently high to cause severe problems. But the sulfate ion is lost through the process. Water that is naturally soft, such as water collected from rain run-off, also contains little or no sulfur, because it has gone through an evaporation-condensation cycle, which leaves behind all the heavier molecules, including sulfur.
Monday, September 27, 2010
2. Sulfur Availability and Obesity Rates
The ultimate source of sulfur is volcanic rock, mainly basalt, spewed up from the earth's core during volcanic eruptions. It is generally believed that humans first evolved from a common ape ancestor in the African rift zone, a region that would have enjoyed an abundance of sulfur due to the heavy volcanic activity there. The three principle suppliers of sulfur to the Western nations are Greece, Italy and Japan. These three countries also enjoy low rates of heart disease and obesity and increased longevity. In South America, a line of volcanoes tracks the backbone of Argentina. Argentinians have a much lower obesity rate than their neighbors to the east in Brazil. In the United States, Oregon and Hawaii, two states with significant volcanic activity, have among the lowest obesity rates in the country. By contrast, the highest obesity rates are found in the midwest and southern farm country: the epicenter of the modern agricultural practices (mega farms) that lead to sulfur depletion in the soil. Among all fifty states, Oregon has the lowest childhood obesity rates. Significantly, Hawaii's youth are faring less well than their parents: while Hawaii ranks as the fifth from the bottom in obesity rates, its children aged 10-17 weigh in at number 13. As Hawaii has recently become increasingly dependent on food imports from the mainland to supply their needs, they have suffered accordingly with increased obesity problems.
3. Why Does Sulfur Deficiency Lead to Obesity?
To summarize what has been said thus far, (1) foods are becoming depleted in sulfur, and (2) locations with naturally high sulfur deposits enjoy protection against obesity. Now comes the difficult question: why does sulfur deficiency lead to obesity? The answer, like much of biology, is complicated, and part of what I theorize is conjecture.
Sulfur is known as a healing mineral, and a sulfur deficiency often leads to pain and inflammation associated with various muscle and skeletal disorders. Sulfur plays a role in many biological processes, one of which is metabolism. Sulfur is present in insulin, the essential hormone that promotes the utilization of sugar derived from carbohydrates for fuel in muscle and fat cells. However, my extensive literature search has led me to two mysterious molecules found in the blood stream and in many other parts of the body: vitamin D3 sulfate and cholesterol sulfate [Strott2003]. Upon exposure to the sun, the skin synthesizes vitamin D3 sulfate, a form of vitamin D that, unlike unsulfated vitamin D3, is water soluble. As a consequence, it can travel freely in the blood stream rather than being packaged up inside LDL (the so-called "bad" cholesterol) for transport [Axelsona1985]. The form of vitamin D that is present in both human milk [Lakdawala1977] and raw cow's milk [Baulch1982] is vitamin D3 sulfate (pasteurization destroys it in cow's milk, and the milk is then artificially enriched with vitamin D2, an unsulfated plant-derived form of the vitamin).
Cholesterol sulfate is also synthesized in the skin, where it forms a crucial part of the barrier that keeps out harmful bacteria and other microorganisms such as fungi [Strott2003]. Cholesterol sulfate regulates the gene for a protein called profilaggrin, by interacting like a hormone with the nuclear receptor ROR-alpha. Profilaggrin is the precursor to filaggrin, which protects the skin from invasive organisms [Sandilands2009, McGrath2008]. A deficiency in filaggrin is associated with asthma and arthritis. Therefore, cholesterol sulfate plays an important role in protection from asthma and arthritis. This explains why sulfur is a healing agent.
Like vitamin D3 sulfate, cholesterol sulfate is also water-soluble, and it too, unlike cholesterol, does not have to be packaged up inside LDL for delivery to the tissues. By the way, vitamin D3 is synthesized through a couple of simple steps from cholesterol, and its chemical structure is, as a consequence, nearly identical to that of cholesterol.
Here I pose the interesting question: where do vitamin D3 sulfate and cholesterol sulfate go once they are in the blood stream, and what role do they play in the cells? Surprisingly, as far as I can tell, nobody knows. It has been determined that the sulfated form of vitamin D3 is strikingly ineffective for calcium transport, the well-known "primary" role of vitamin D3 [Reeve1981]. However, vitamin D3 clearly has many other positive effects (it seems that more and more are being discovered every day), and these include a role in cancer protection, increased immunity against infectious disease, and protection against heart disease ( Vitamin D Protects against Cancer and Autoimmune Diseases). Researchers don't yet understand how it achieves these benefits, which have been observed empirically but remain unexplained physiologically. However, I strongly suspect it is the sulfated form of the vitamin that instantiates these benefits, and my reasons for this belief will become clearer in a moment.
One very special feature of cholesterol sulfate, as opposed to cholesterol itself, is that it is very agile: due to its polarity it can freely pass through cell membranes almost like a ghost [Rodriguez1995]. This means that cholesterol sulfate can easily enter a fat or muscle cell. I am developing a theory which at its core proposes an essential role for cholesterol sulfate in the metabolism of glucose for fuel by these cells. Below, I will show how cholesterol sulfate may be able to protect fat and muscle cells from damage due to exposure to glucose, a dangerous reducing agent, and to oxygen, a dangerous oxidizing agent. I will further argue that, with insufficient cholesterol sulfate, muscle and fat cells become damaged, and as a consequence become glucose intolerant: unable to process glucose as a fuel. This happens first to muscle cells but eventually to fat cells, as well. Fat cells become storage bins for fats to supply fuel to the muscles, because the muscles are unable to utilize glucose as fuel. Eventually, fat cells also become too disabled to release their stored fats. Fatty tissue then accumulates on the body.
Sulfur is known as a healing mineral, and a sulfur deficiency often leads to pain and inflammation associated with various muscle and skeletal disorders. Sulfur plays a role in many biological processes, one of which is metabolism. Sulfur is present in insulin, the essential hormone that promotes the utilization of sugar derived from carbohydrates for fuel in muscle and fat cells. However, my extensive literature search has led me to two mysterious molecules found in the blood stream and in many other parts of the body: vitamin D3 sulfate and cholesterol sulfate [Strott2003]. Upon exposure to the sun, the skin synthesizes vitamin D3 sulfate, a form of vitamin D that, unlike unsulfated vitamin D3, is water soluble. As a consequence, it can travel freely in the blood stream rather than being packaged up inside LDL (the so-called "bad" cholesterol) for transport [Axelsona1985]. The form of vitamin D that is present in both human milk [Lakdawala1977] and raw cow's milk [Baulch1982] is vitamin D3 sulfate (pasteurization destroys it in cow's milk, and the milk is then artificially enriched with vitamin D2, an unsulfated plant-derived form of the vitamin).
Cholesterol sulfate is also synthesized in the skin, where it forms a crucial part of the barrier that keeps out harmful bacteria and other microorganisms such as fungi [Strott2003]. Cholesterol sulfate regulates the gene for a protein called profilaggrin, by interacting like a hormone with the nuclear receptor ROR-alpha. Profilaggrin is the precursor to filaggrin, which protects the skin from invasive organisms [Sandilands2009, McGrath2008]. A deficiency in filaggrin is associated with asthma and arthritis. Therefore, cholesterol sulfate plays an important role in protection from asthma and arthritis. This explains why sulfur is a healing agent.
Like vitamin D3 sulfate, cholesterol sulfate is also water-soluble, and it too, unlike cholesterol, does not have to be packaged up inside LDL for delivery to the tissues. By the way, vitamin D3 is synthesized through a couple of simple steps from cholesterol, and its chemical structure is, as a consequence, nearly identical to that of cholesterol.
Here I pose the interesting question: where do vitamin D3 sulfate and cholesterol sulfate go once they are in the blood stream, and what role do they play in the cells? Surprisingly, as far as I can tell, nobody knows. It has been determined that the sulfated form of vitamin D3 is strikingly ineffective for calcium transport, the well-known "primary" role of vitamin D3 [Reeve1981]. However, vitamin D3 clearly has many other positive effects (it seems that more and more are being discovered every day), and these include a role in cancer protection, increased immunity against infectious disease, and protection against heart disease ( Vitamin D Protects against Cancer and Autoimmune Diseases). Researchers don't yet understand how it achieves these benefits, which have been observed empirically but remain unexplained physiologically. However, I strongly suspect it is the sulfated form of the vitamin that instantiates these benefits, and my reasons for this belief will become clearer in a moment.
One very special feature of cholesterol sulfate, as opposed to cholesterol itself, is that it is very agile: due to its polarity it can freely pass through cell membranes almost like a ghost [Rodriguez1995]. This means that cholesterol sulfate can easily enter a fat or muscle cell. I am developing a theory which at its core proposes an essential role for cholesterol sulfate in the metabolism of glucose for fuel by these cells. Below, I will show how cholesterol sulfate may be able to protect fat and muscle cells from damage due to exposure to glucose, a dangerous reducing agent, and to oxygen, a dangerous oxidizing agent. I will further argue that, with insufficient cholesterol sulfate, muscle and fat cells become damaged, and as a consequence become glucose intolerant: unable to process glucose as a fuel. This happens first to muscle cells but eventually to fat cells, as well. Fat cells become storage bins for fats to supply fuel to the muscles, because the muscles are unable to utilize glucose as fuel. Eventually, fat cells also become too disabled to release their stored fats. Fatty tissue then accumulates on the body.
4. Sulfur and Glucose Metabolism
In order to understand my theory, you will need to know more about glucose metabolism. Skeletal muscle cells and fat cells break down glucose in the presence of oxygen in their mitochondria, and in the process they produce ATP, the basic energy currency of all cells. A glucose transporter called GLUT4 is present in the cytoplasm of muscle cells, and it migrates to the cell membrane upon stimulation by insulin. GLUT4 essentially acts as a key that unlocks the door, letting glucose into the cell, but, like a key, it only works when it's inserted in the membrane. Both glucose and oxygen, unless they are carefully managed, can cause harm to the cell's proteins and fats. The glucose enters the cell within special cholesterol rich sites in the cell wall called lipid rafts [Inoue2006]. This is likely orchestrated to protect the cell wall from damage, because extra cholesterol allows the vulnerable lipoproteins in the cell wall to pack more tightly and reduce their risk of exposure. In muscle cells, myoglobin is able to store additional oxygen, bound to an iron molecule safely sequestered in an interior cavity within the myoglobin protein.
Sulfur is a very versatile molecule, because it can exist in several distinct oxidative states, ranging from +6 (in the sulfate radical) to -2 (in hydrogen sulfide). Glucose, as a powerful reducing agent, can cause significant glycation damage to exposed proteins, leading to the formation of Advanced Glycation End Products (AGE's) that are extremely destructive to health: they are believed to be a major contributor to heart disease risk [Brownlee1988]. So, I hypothesize that, if sulfur (+6) is made available to glucose as a decoy, the glucose will be diverted into reducing the sulfur rather than glycating some vulnerable protein such as myoglobin.
In searching the Web, I came across an article written in the 1930's about the striking ability of iron sulfate, in the presence of the oxidizing agent hydrogen peroxide, to break down starch into simple molecules, even in the absence of any enzymes to catalyze the reaction [Brown1936]. The article pointedly mentioned that iron works much better than other metals, and sulfate works much better than other anions. In the human body, starch is first converted to glucose in the digestive system. The muscle and fat cells only need to break down glucose. Thus, their task is easier, because the iron sulfate is now starting from an intermediate breakdown product of starch rather than from starch itself.
Where would the iron sulfate come from? It seems to me that the cholesterol sulfate, having hopped across the cell membrane, could transfer its sulfate radical to the myoglobin, whose iron molecule could provide the other half of the formula. In the process, the sulfur molecule's charge would be driven down from +6 to -2, releasing energy and absorbing the impact of the reducing effects of glucose, and therefore serving as a decoy to protect the proteins in the cell from glycation damage.
When the cell is exposed to insulin, its mitochondria are triggered to start pumping both hydrogen peroxide and hydrogen ions into the cytoplasm, essentially gearing up for the assault by glucose. If cholesterol sulfate enters the cell alongside the glucose, then all the players are available. I conjecture that cholesterol sulfate is the catalyst that seeds the lipid raft. Iron sulfate is then formed by bonding the iron in the heme unit in myoglobin to a sulfate ion provided by cholesterol sulfate. The cholesterol is left behind in the cell wall, thus enriching the newly forming lipid raft with cholesterol. The hydrogen peroxide, provided by the mitochondria upon insulin stimulation, catalyzes the dissolution of glucose by the iron sulfate. The pumped hydrogen can pair up with the reduced sulfur (S-2) to form hydrogen sulfide, a gas that can easily diffuse back across the membrane for a repeat cycle. The oxygen that is released from the sulfate radical is picked up by the myoglobin, sequestered inside the molecule for safe travel to the mitochondria. Glucose breakdown products and oxygen are then delivered to the mitochondria to complete the process that ends with water, carbon dioxide, and ATP -- all while keeping the cell's cytoplasmic proteins safe from glucose and oxygen exposure.
If I'm right about this role for cholesterol sulfate both in seeding the lipid raft and in providing the sulfate ion, then this process breaks down miserably when cholesterol sulfate is not available. First of all, the lipid raft is not formed. Without the lipid raft, the glucose can not enter the cell. Intense physical exercise can allow glucose to enter the muscle cells even in the absence of insulin [Ojuka2002]. However, this will lead to dangerous exposure of the cell's proteins to glycation (because there is no iron sulfate to degrade the glucose). Glycation interferes with the proteins' ability to perform their jobs, and leaves them more vulnerable to oxidation damage. One of the important affected proteins would be myoglobin: it would no longer be able to effectively carry oxygen to the mitochondria. Furthermore, oxidized myoglobin released into the blood stream by crippled muscle cells leads to painful and crippling rhabdomyolysis, and possible subsequent kidney failure. This explanation accounts for the observation that sulfur deficiency leads to muscle pain and inflammation.
Sulfur is a very versatile molecule, because it can exist in several distinct oxidative states, ranging from +6 (in the sulfate radical) to -2 (in hydrogen sulfide). Glucose, as a powerful reducing agent, can cause significant glycation damage to exposed proteins, leading to the formation of Advanced Glycation End Products (AGE's) that are extremely destructive to health: they are believed to be a major contributor to heart disease risk [Brownlee1988]. So, I hypothesize that, if sulfur (+6) is made available to glucose as a decoy, the glucose will be diverted into reducing the sulfur rather than glycating some vulnerable protein such as myoglobin.
In searching the Web, I came across an article written in the 1930's about the striking ability of iron sulfate, in the presence of the oxidizing agent hydrogen peroxide, to break down starch into simple molecules, even in the absence of any enzymes to catalyze the reaction [Brown1936]. The article pointedly mentioned that iron works much better than other metals, and sulfate works much better than other anions. In the human body, starch is first converted to glucose in the digestive system. The muscle and fat cells only need to break down glucose. Thus, their task is easier, because the iron sulfate is now starting from an intermediate breakdown product of starch rather than from starch itself.
Where would the iron sulfate come from? It seems to me that the cholesterol sulfate, having hopped across the cell membrane, could transfer its sulfate radical to the myoglobin, whose iron molecule could provide the other half of the formula. In the process, the sulfur molecule's charge would be driven down from +6 to -2, releasing energy and absorbing the impact of the reducing effects of glucose, and therefore serving as a decoy to protect the proteins in the cell from glycation damage.
When the cell is exposed to insulin, its mitochondria are triggered to start pumping both hydrogen peroxide and hydrogen ions into the cytoplasm, essentially gearing up for the assault by glucose. If cholesterol sulfate enters the cell alongside the glucose, then all the players are available. I conjecture that cholesterol sulfate is the catalyst that seeds the lipid raft. Iron sulfate is then formed by bonding the iron in the heme unit in myoglobin to a sulfate ion provided by cholesterol sulfate. The cholesterol is left behind in the cell wall, thus enriching the newly forming lipid raft with cholesterol. The hydrogen peroxide, provided by the mitochondria upon insulin stimulation, catalyzes the dissolution of glucose by the iron sulfate. The pumped hydrogen can pair up with the reduced sulfur (S-2) to form hydrogen sulfide, a gas that can easily diffuse back across the membrane for a repeat cycle. The oxygen that is released from the sulfate radical is picked up by the myoglobin, sequestered inside the molecule for safe travel to the mitochondria. Glucose breakdown products and oxygen are then delivered to the mitochondria to complete the process that ends with water, carbon dioxide, and ATP -- all while keeping the cell's cytoplasmic proteins safe from glucose and oxygen exposure.
If I'm right about this role for cholesterol sulfate both in seeding the lipid raft and in providing the sulfate ion, then this process breaks down miserably when cholesterol sulfate is not available. First of all, the lipid raft is not formed. Without the lipid raft, the glucose can not enter the cell. Intense physical exercise can allow glucose to enter the muscle cells even in the absence of insulin [Ojuka2002]. However, this will lead to dangerous exposure of the cell's proteins to glycation (because there is no iron sulfate to degrade the glucose). Glycation interferes with the proteins' ability to perform their jobs, and leaves them more vulnerable to oxidation damage. One of the important affected proteins would be myoglobin: it would no longer be able to effectively carry oxygen to the mitochondria. Furthermore, oxidized myoglobin released into the blood stream by crippled muscle cells leads to painful and crippling rhabdomyolysis, and possible subsequent kidney failure. This explanation accounts for the observation that sulfur deficiency leads to muscle pain and inflammation.
5. The Metabolc Syndrome
The metabolic syndrome is a term used to encapsulate a complex set of markers associated with increased risk to heart disease. The profile includes (1) insulin resistance and dysfunctional glucose metabolism in muscle cells, (2) excess triglycerides in the blood serum, (3) high levels of LDL, particularly small dense LDL, the worst kind, (4) low levels of HDL (the "good" cholesterol) and reduced cholesterol content within the individual HDL particles, (5) elevated blood pressure, and (6) obesity, particularly excess abdominal fat. I have argued previously that this syndrome is brought on by a diet that is high in empty carbohydrates (particularly fructose) and low in fats and cholesterol, along with a poor vitamin D status [Seneff2010]. While I still believe that all of these factors are contributory, I would now add another factor as well: insufficient dietary sulfate.
I have described in a previous essay, my interpretation of obesity as being driven by a need for abundant fat cells to convert glucose to fat because the muscle cells are unable to efficiently utilize glucose as fuel. With sulfur deficiency comes the answer as to why muscle cells would be defective in glucose management: they can't come up with enough cholesterol sulfate to seed the lipid raft needed to import the glucose.
An alternative way to ovecome a muscle cell's defective glucose metabolism is to exercise vigorously, so that the generated AMPK (an indicator of energy shortage) induces the GLUT4 to migrate to the membrane even in the absence of insulin [Ojuka2002]. Once the glucose is inside the muscle cell, however, the iron-sulfate mechanism just described is dysfunctional, both because there's no cholesterol sulfate and because there's no hydrogen peroxide. Additionally, with intensive exercise there's also a reduced supply of oxygen, so the glucose must be processed anaerobically in the cytoplasm to produce lactate. The lactate is released into the blood stream and shipped to the heart and brain, both of which are able to use it as fuel. But the cell membrane remains depleted in cholesterol, and this makes it vulnerable to future oxidative damage.
Another way to compensate for defective glucose metabolism in the muscle cells is to gain weight. Fat cells must now convert glucose into fat and release it into the blood stream as triglycerides, to fuel the muscle cells. In the context of a low fat diet, sulfur deficiency becomes that much worse a problem. Sulfur deficiency interferes with glucose metabolism, so it's a much healthier choice to simply avoid glucose sources (carbohydrates) in the diet; i.e. to adopt a very low-carb diet. Then the fat in the diet can supply the muscles with fuel, and the fat cells are not burdened with having to store up so much reserve fat.
Insulin suppresses the release of fats from fat cells [Scappola1995]. This forces the fat cells to flood the bloodstream with triglycerides when insulin levels are low, i.e., after prolonged periods of fasting, such as overnight. The fat cells must dump enough triglycerides into the bloodstream during fasting periods to fuel the muscles when the dietary supply of carbohydrates keeps insulin levels elevated, and the release of fats from the fat cells is repressed. As the dietary carbs come in, blood sugar levels rise dramatically because the muscle cells can't utilize it.
The liver also processes excess glucose into fat, and packages it up into LDL, to further supply fuel to the defective muscle cells. Because the liver is so preoccupied with processing glucose and fructose into LDL, it falls behind on the generation of HDL, the "good" cholesterol. So the result is elevated levels of LDL, triglycerides, and blood sugar, and reduced levels of HDL, four key components of the metabolic syndrome.
The chronic presence of excess glucose and fructose in the blood stream leads to a host of problems, all related to glycation damage of blood stream proteins by glucose exposure. One of the key proteins that gets damaged is the apolipoprotein, apoB, that's encased in the membrane of the LDL particles. Damaged apoB inhibits the ability of LDL to efficiently deliver its contents (fat and cholesterol) to the tissues. Fat cells again come to the rescue, by scavenging the broken LDL particles (through a mechanism that does not require apoB to be healthy), taking them apart, and extracting and refurbishing their cholesterol. In order to function properly, the fat cells must have intact ApoE, an antioxidant that cleans up oxidized cholesterol and transports it to the cell membrane for delivery to HDL particles.
I have described in a previous essay, my interpretation of obesity as being driven by a need for abundant fat cells to convert glucose to fat because the muscle cells are unable to efficiently utilize glucose as fuel. With sulfur deficiency comes the answer as to why muscle cells would be defective in glucose management: they can't come up with enough cholesterol sulfate to seed the lipid raft needed to import the glucose.
An alternative way to ovecome a muscle cell's defective glucose metabolism is to exercise vigorously, so that the generated AMPK (an indicator of energy shortage) induces the GLUT4 to migrate to the membrane even in the absence of insulin [Ojuka2002]. Once the glucose is inside the muscle cell, however, the iron-sulfate mechanism just described is dysfunctional, both because there's no cholesterol sulfate and because there's no hydrogen peroxide. Additionally, with intensive exercise there's also a reduced supply of oxygen, so the glucose must be processed anaerobically in the cytoplasm to produce lactate. The lactate is released into the blood stream and shipped to the heart and brain, both of which are able to use it as fuel. But the cell membrane remains depleted in cholesterol, and this makes it vulnerable to future oxidative damage.
Another way to compensate for defective glucose metabolism in the muscle cells is to gain weight. Fat cells must now convert glucose into fat and release it into the blood stream as triglycerides, to fuel the muscle cells. In the context of a low fat diet, sulfur deficiency becomes that much worse a problem. Sulfur deficiency interferes with glucose metabolism, so it's a much healthier choice to simply avoid glucose sources (carbohydrates) in the diet; i.e. to adopt a very low-carb diet. Then the fat in the diet can supply the muscles with fuel, and the fat cells are not burdened with having to store up so much reserve fat.
Insulin suppresses the release of fats from fat cells [Scappola1995]. This forces the fat cells to flood the bloodstream with triglycerides when insulin levels are low, i.e., after prolonged periods of fasting, such as overnight. The fat cells must dump enough triglycerides into the bloodstream during fasting periods to fuel the muscles when the dietary supply of carbohydrates keeps insulin levels elevated, and the release of fats from the fat cells is repressed. As the dietary carbs come in, blood sugar levels rise dramatically because the muscle cells can't utilize it.
The liver also processes excess glucose into fat, and packages it up into LDL, to further supply fuel to the defective muscle cells. Because the liver is so preoccupied with processing glucose and fructose into LDL, it falls behind on the generation of HDL, the "good" cholesterol. So the result is elevated levels of LDL, triglycerides, and blood sugar, and reduced levels of HDL, four key components of the metabolic syndrome.
The chronic presence of excess glucose and fructose in the blood stream leads to a host of problems, all related to glycation damage of blood stream proteins by glucose exposure. One of the key proteins that gets damaged is the apolipoprotein, apoB, that's encased in the membrane of the LDL particles. Damaged apoB inhibits the ability of LDL to efficiently deliver its contents (fat and cholesterol) to the tissues. Fat cells again come to the rescue, by scavenging the broken LDL particles (through a mechanism that does not require apoB to be healthy), taking them apart, and extracting and refurbishing their cholesterol. In order to function properly, the fat cells must have intact ApoE, an antioxidant that cleans up oxidized cholesterol and transports it to the cell membrane for delivery to HDL particles.
6. Fat Cells, Macrophages and Atherosclerosis
While diligently converting glucose to stored fats, the fat cells are awash in glucose, which damages their apoE through glycation [Li1997]. Once their apoE is damaged, they can no longer transport cholesterol to the membrane. Excess cholesterol accumulates inside the fat cells and eventually destroys their ability to synthesize proteins. Concurrently, their cell membrane becomes depleted in cholesterol, because they can no longer deliver it to the membrane [Seneff2010]. A fat cell that has deteriorated to this degree has no choice but to die: it sends out distress signals that call in macrophages. The macrophages essentially consume the dysfunctional fat cell, wrapping their own membrane around the fat cell's membrane that is now barely able to hold its contents inside [Cinti2005].
Macrophages are also principle players in the fatty streaks that appear along the sides of major arteries leading to the heart, and are associated with plaque build-up and heart disease. In a fascinating set of experiments, Ma et al. [Ma2008] have shown that the sulfate ion attached to oxidized forms of cholesterol is highly protective against fatty streaks and atherosclerosis. In a set of in-vitro experiments, they demonstrated diametrically opposite reactions from macrophages to 25-hydroxyl cholesterol (25-HC) versus its sulfoconjugate 25-hydroxyl cholesterol sulfate (25-HC3S). Whereas 25-HC present in the medium causes the macrophages to synthesize and store cholesterol and fatty acids, 25-HC3S has the exact opposite effect: it promotes the release of cholesterol to the medium and causes fat stores to shrink. Furthermore, while 25-HC added to the medium led to apoptosis and cell death, 25-HC3S did not. I suggest that the sulfate radical is essential for the process that feeds cholesterol and oxygen to the heart muscle.
Macrophages are also principle players in the fatty streaks that appear along the sides of major arteries leading to the heart, and are associated with plaque build-up and heart disease. In a fascinating set of experiments, Ma et al. [Ma2008] have shown that the sulfate ion attached to oxidized forms of cholesterol is highly protective against fatty streaks and atherosclerosis. In a set of in-vitro experiments, they demonstrated diametrically opposite reactions from macrophages to 25-hydroxyl cholesterol (25-HC) versus its sulfoconjugate 25-hydroxyl cholesterol sulfate (25-HC3S). Whereas 25-HC present in the medium causes the macrophages to synthesize and store cholesterol and fatty acids, 25-HC3S has the exact opposite effect: it promotes the release of cholesterol to the medium and causes fat stores to shrink. Furthermore, while 25-HC added to the medium led to apoptosis and cell death, 25-HC3S did not. I suggest that the sulfate radical is essential for the process that feeds cholesterol and oxygen to the heart muscle.
7. Sulfur and Alzheimer's
With an aging population, Alzheimer's disease is on the rise, and it has been argued that the rate of increase is disproportionately high compared to the increase in the raw number of elderly people [Waldman2009]. Because of a conviction that the amyloid beta plaque that is a signature of Alzheimer's is also the cause, the pharmaceutical industry has spent hundreds of millions, if not billions, of dollars pursuing drugs that reduce the amount of plaque accumulating in the brain. Thus far, drug trials have been so disappointing that many are beginning to believe that amyloid beta is not the cause after all. Recent drug trials have shown not only no improvement, but actually a further decline in cognitive function, compared to placebo ( New York Times Article). I have argued elsewhere that amyloid beta may actually be protective against Alzheimer's, and that problems with glucose metabolism are the true culprit in the disease.
Once I began to suspect sulfur deficiency as a major factor in Americans' health, I looked into the relationship between sulfur deficiency and Alzheimer's. Imagine my surprise when I came upon a web page posted by Ronald Roth, which shows a plot of the levels of various minerals in the cells of a typical Alzheimer's patient relative to the normal level. Remarkably, sulfur is almost non-existent in the Alzheimer's patient's profile.
To quote directly from that site: "While some drugs or antibiotics may slow, or if it should happen, halt the progression of Alzheimer's disease, sulfur supplementation has the potential of not only preventing, but actually reversing the condition, provided it has not progressed to a stage where much damage has been done to the brain."
"One major reason for the increase in Alzheimer's disease over the past years has been the bad reputation eggs have been getting in respect to being a high source of cholesterol, despite the fact of dietary intake of cholesterol having little impact on serum cholesterol - which is now also finally acknowledged by mainstream medicine. In the meantime, a large percentage of the population lost out on an excellent source of sulfur and a host of other essential nutrients by following the nutritional misinformation spread on eggs. Of course, onions and garlic are another rich source of sulfur, but volume-wise, they cannot duplicate the amounts obtained from regularly consuming eggs."
Why should sulfur deficiency be so important for the brain? I suspect that the answer lies in the mysterious molecule alpha-synuclein, which shows up alongside amyloid-beta in the plaque, and is also present in the Lewy Bodies that are a signature of Parkinson's disease [Olivares2009]. The alpha-synuclein molecule contains four methionine residues, and all four of the sulfur molecules in the methionine residues are converted to sulfoxides in the presence of oxidizing agents such as hydrogen peroxide [Glaser2005]. Just as in the muscle cells, insulin would cause the mitochondria of neurons to release hydrogen peroxide, which would then allow the alpha-synuclein to take up oxygen, in a way that is very reminiscent of what myoglobin can do in muscle cells. The lack of sufficient sulfur should directly impact the neuron's ability to safely carry oxygen, again paralleling the situation in muscle cells. This would mean that other proteins and fats in the neuron would suffer from oxidative damage, leading ultimately to the neuron's destruction.
In my essay on Alzheimer's, I argued that biologically pro-active restriction in glucose metabolism in the brain (a so-called type-III diabetes and a precursor to Alzheimer's disease) is triggered by a deficiency in cholesterol in the neuron cell membrane. Again, as in muscle cells, glucose entry depends upon cholesterol-rich lipid rafts, and, when the cell is deficient in cholesterol, the brain goes into a mode of metabolism that prefers other nutrients besides glucose.
I suspect that a deficiency in cholesterol would come about if there is insufficient cholesterol sulfate, because cholesterol sulfate likely plays an important role in seeding lipid rafts, while concurrently enriching the cell wall in cholesterol. The cell also develops an insensitivity to insulin, and, as a consequence, anaerobic metabolism becomes favored over aerobic metabolism, reducing the chances for alpha-synuclein to become oxidized. Oxidation actually protects alpha-synuclein from fibrillation, a necessary structural change for the accumulation of Lewy bodies in Parkinson's disease (and likely also Alzheimer's plaque) [Glaser2005]
Once I began to suspect sulfur deficiency as a major factor in Americans' health, I looked into the relationship between sulfur deficiency and Alzheimer's. Imagine my surprise when I came upon a web page posted by Ronald Roth, which shows a plot of the levels of various minerals in the cells of a typical Alzheimer's patient relative to the normal level. Remarkably, sulfur is almost non-existent in the Alzheimer's patient's profile.
To quote directly from that site: "While some drugs or antibiotics may slow, or if it should happen, halt the progression of Alzheimer's disease, sulfur supplementation has the potential of not only preventing, but actually reversing the condition, provided it has not progressed to a stage where much damage has been done to the brain."
"One major reason for the increase in Alzheimer's disease over the past years has been the bad reputation eggs have been getting in respect to being a high source of cholesterol, despite the fact of dietary intake of cholesterol having little impact on serum cholesterol - which is now also finally acknowledged by mainstream medicine. In the meantime, a large percentage of the population lost out on an excellent source of sulfur and a host of other essential nutrients by following the nutritional misinformation spread on eggs. Of course, onions and garlic are another rich source of sulfur, but volume-wise, they cannot duplicate the amounts obtained from regularly consuming eggs."
Why should sulfur deficiency be so important for the brain? I suspect that the answer lies in the mysterious molecule alpha-synuclein, which shows up alongside amyloid-beta in the plaque, and is also present in the Lewy Bodies that are a signature of Parkinson's disease [Olivares2009]. The alpha-synuclein molecule contains four methionine residues, and all four of the sulfur molecules in the methionine residues are converted to sulfoxides in the presence of oxidizing agents such as hydrogen peroxide [Glaser2005]. Just as in the muscle cells, insulin would cause the mitochondria of neurons to release hydrogen peroxide, which would then allow the alpha-synuclein to take up oxygen, in a way that is very reminiscent of what myoglobin can do in muscle cells. The lack of sufficient sulfur should directly impact the neuron's ability to safely carry oxygen, again paralleling the situation in muscle cells. This would mean that other proteins and fats in the neuron would suffer from oxidative damage, leading ultimately to the neuron's destruction.
In my essay on Alzheimer's, I argued that biologically pro-active restriction in glucose metabolism in the brain (a so-called type-III diabetes and a precursor to Alzheimer's disease) is triggered by a deficiency in cholesterol in the neuron cell membrane. Again, as in muscle cells, glucose entry depends upon cholesterol-rich lipid rafts, and, when the cell is deficient in cholesterol, the brain goes into a mode of metabolism that prefers other nutrients besides glucose.
I suspect that a deficiency in cholesterol would come about if there is insufficient cholesterol sulfate, because cholesterol sulfate likely plays an important role in seeding lipid rafts, while concurrently enriching the cell wall in cholesterol. The cell also develops an insensitivity to insulin, and, as a consequence, anaerobic metabolism becomes favored over aerobic metabolism, reducing the chances for alpha-synuclein to become oxidized. Oxidation actually protects alpha-synuclein from fibrillation, a necessary structural change for the accumulation of Lewy bodies in Parkinson's disease (and likely also Alzheimer's plaque) [Glaser2005]
8. Is The Skin a Solar-Powered Battery for the Heart?
The evidence is quite compelling that sunny places afford protection from heart disease. A study described in [Grimes1996] provides an in depth anaylsis of data from around the world showing an inverse relationship between heart disease rates and sunny climate/low latitude. For instance, the cardiovascular-related death rate for men between the ages of 55 and 64 was 761 per 100,000 men in Belfast, Northern Ireland, but only 175 in Toulouse, France. While the obvious biological factor that would be impacted by sunlight is vitamin D, studies conducted specifically on vitamin D status have been inconclusive, with some even showing a significant increased risk for heart disease with increased intake of vitamin D2 supplements [Drolet2003].
I believe, first of all, that the distinction between vitamin D3 and vitamin D3-sulfate really matters, and also that the distinction between vitamin D2 and vitamin D3 really matters. Vitamin D2 is the plant form of the vitamin -- it works similarly to D3 with respect to calcium transport, but it cannot be sulfated. Furthermore, apparently the body is unable to produce vitamin D3 sulfate directly from unsulfated vitamin D3 [Lakdawala1977] (which implies that it produces vitamin D3 sulfate directly from cholesterol sulfate). I am not aware of any other food source besides raw milk that contains vitamin D3 in the sulfated form. So, when studies monitor either vitamin D supplements or vitamin D serum levels, they're not getting at the crucial aspect for heart protection, which I think is the serum level of vitamin D3 sulfate.
Furthermore, I believe it is extremely likely that vitamin D3 sulfate is not the only thing that's impacted by greater sun exposure, and maybe not even the most important thing. Given that cholesterol sulfate and vitamin D3 sulfate are very similar in molecular structure, I would imagine that both molecules are produced the same way. And since vitamin D3-sulfate synthesis requires sun exposure, I suspect that cholesterol sulfate synthesis may also exploit the sun's radiation energy.
Both cholesterol and sulfur afford protection in the skin from radiation damage to the cell's DNA, the kind of damage that can lead to skin cancer. Cholesterol and sulfur become oxidized upon exposure to the high frequency rays in sunlight, thus acting as antioxidants to "take the heat," so to speak. Oxidation of cholesterol is the first step in the process by which cholesterol transforms itself into vitamin D3. Sulfur dioxide in the air is converted nonenzymatically to the sulfate ion upon sun exposure. This is the process that produces acid rain. The oxidation of sulfide (S-2) to sulfate (SO4-2), a strongly endothermic reaction [Hockin2003], converts the sun's energy into chemical energy contained in the sulfur-oxygen bonds, while simultaneously picking up four oxygen molecules. Attaching the sulfate ion to cholesterol or vitamin D3 is an ingenious step, because it makes these molecules water-soluble and therefore easily transportable through the blood stream.
Hydrogen sulfide (H2S) is consistently found in the blood stream in small amounts. As a gas, it can diffuse into the air from capillaries close to the skin's surface. So it is conceivable that we rely on bacteria in the skin to convert sulfide to sulfate. It would not be the first time that humans have struck up a symbiotic relationship with bacteria. If this is true, then washing the skin with antibiotic soap is a bad idea. Phototrophic bacteria, such as Chlorobium tepidum, that can convert H2S to H2SO4 exist in nature [Zerkle2009, Wahlund1991], for example in sulfur hot springs in Yellowstone Park. These highly specialized bacteria can convert the light energy from the sun into chemical energy in the sulfate ion.
Another possibility is that we have specialized cells in the skin, possibly the keratinocytes, that are able to exploit sunlight to convert sulfide to sulfate, using a similar phototrophic mechanism to C. tepidum. This seems quite plausible, especially considering that both human keratinocytes and C. tepidum can synthesize an interesting UV-B absorbing cofactor, tetrahydrobioptin. This cofactor is found universally in mammalian cells, and one of its roles is to regulate the synthesis of melanin [Schallreut94], the skin pigment that is associated with a tan and protects the skin from damage by UV-light exposure [Costin2007]. However, tetrahydrobiopsin is very rare in the bacterial kingdom, and C. tepidum is one of the very few bacteria that can synthesize it [Cho99].
Let me summarize at this point where I'm on solid ground and where I'm speculating. It is undisputed that the skin synthesizes cholesterol sulfate in large amounts, and it has been suggested that the skin is the major supplier of cholesterol sulfate to the blood stream [Strott2003]. The skin also synthesizes vitamin D3 sulfate, upon exposure to sunlight. Vitamin D3 is synthesized from cholesterol, with oxysterols (created from sun exposure) as an intermediate step (oxysterols are forms of cholesterol with hydroxyl groups attached at various places in the carbon chain). The body can't synthesize vitamin D3 sulfate from vitamin D3 [Lakdawala1977] so it must be that sulfation happens first, producing cholesterol sulfate or hydroxy-cholesterol sulfate, which is then optionally converted to vitamin D3 sulfate or shipped out "as is."
Another highly significant feature of skin cells is that the skin stores sulfate ions attached to molecules that are universally present in the intracellular matrix, such as heparan sulfate, chondroitin sulfate, and keratin sulfate [Milstone1994]. Furthermore, it has been shown that exposure of the melanin producing cells (melanocytes) to molecules containing reduced sulfur (-2) leads to suppression of melanin synthesis [Chu2009], whereas exposure to molecules like chondroitin sulfate that contain oxidized sulfur (+6) leads to enhancement of melanin synthesis [Katz1976]. Melanin is a potent UV-light absorber, and it would compete with reduced sulfur for the opportunity to become oxidized. It is therefore logical that, when sulfur is reduced, melanin synthesis should be suppressed, so that sulfur can absorb the solar energy and convert it to very useful chemical bonds in the sulfate ion.
The sulfate would eventually be converted back to sulfide by a muscle cell in the heart or a skeletal muscle (simultaneously recovering the energy to fuel the cell and unlocking the oxygen to support aerobic metabolism of glucose), and the cycle would continually repeat.
Why am I spending so much time talking about all of this? Well, if I'm right, then the skin can be viewed as a solar-powered battery for the heart, and that is a remarkable concept. The energy in sunlight is converted into chemical energy in the oxygen-sulfur bonds, and then transported through the blood vessels to the heart and skeletal muscles. The cholesterol sulfate and vitamin D3 sufate are carriers that deliver the energy (and the oxygen) "door-to-door" to the individual heart and skeletal muscle cells.
Today's lifestyle, especially in America, severely stresses this system. First of all, most Americans believe that any food containing cholesterol is unhealthy, so the diet is extremely low in cholesterol. Eggs are an excellent source of sulfur, but because of their high cholesterol content we have been advised to eat them sparingly. Secondly, as I discussed previously, natural food plant sources of sulfur are likely to be deficient due to sulfur depletion in the soil. Thirdly, water softeners remove sulfur from our water supply, which would otherwise be a good source. Fourthly, we have been discouraged from eating too much red meat, an excellent source of sulfur-containing amino acids. Finally, we have been instructed by doctors and other authoritarian sources to stay out of the sun and wear high SPF sunscreen whenever we do get sun exposure.
Another significant contributor is the high carbohydrate, low fat diet, which leads to excess glucose in the blood stream that glycates LDL particles and renders them ineffective in delivering cholesterol to the tissues. One of those tissues is the skin, so skin becomes further depleted in cholesterol due to glycation damage to LDL.
I believe, first of all, that the distinction between vitamin D3 and vitamin D3-sulfate really matters, and also that the distinction between vitamin D2 and vitamin D3 really matters. Vitamin D2 is the plant form of the vitamin -- it works similarly to D3 with respect to calcium transport, but it cannot be sulfated. Furthermore, apparently the body is unable to produce vitamin D3 sulfate directly from unsulfated vitamin D3 [Lakdawala1977] (which implies that it produces vitamin D3 sulfate directly from cholesterol sulfate). I am not aware of any other food source besides raw milk that contains vitamin D3 in the sulfated form. So, when studies monitor either vitamin D supplements or vitamin D serum levels, they're not getting at the crucial aspect for heart protection, which I think is the serum level of vitamin D3 sulfate.
Furthermore, I believe it is extremely likely that vitamin D3 sulfate is not the only thing that's impacted by greater sun exposure, and maybe not even the most important thing. Given that cholesterol sulfate and vitamin D3 sulfate are very similar in molecular structure, I would imagine that both molecules are produced the same way. And since vitamin D3-sulfate synthesis requires sun exposure, I suspect that cholesterol sulfate synthesis may also exploit the sun's radiation energy.
Both cholesterol and sulfur afford protection in the skin from radiation damage to the cell's DNA, the kind of damage that can lead to skin cancer. Cholesterol and sulfur become oxidized upon exposure to the high frequency rays in sunlight, thus acting as antioxidants to "take the heat," so to speak. Oxidation of cholesterol is the first step in the process by which cholesterol transforms itself into vitamin D3. Sulfur dioxide in the air is converted nonenzymatically to the sulfate ion upon sun exposure. This is the process that produces acid rain. The oxidation of sulfide (S-2) to sulfate (SO4-2), a strongly endothermic reaction [Hockin2003], converts the sun's energy into chemical energy contained in the sulfur-oxygen bonds, while simultaneously picking up four oxygen molecules. Attaching the sulfate ion to cholesterol or vitamin D3 is an ingenious step, because it makes these molecules water-soluble and therefore easily transportable through the blood stream.
Hydrogen sulfide (H2S) is consistently found in the blood stream in small amounts. As a gas, it can diffuse into the air from capillaries close to the skin's surface. So it is conceivable that we rely on bacteria in the skin to convert sulfide to sulfate. It would not be the first time that humans have struck up a symbiotic relationship with bacteria. If this is true, then washing the skin with antibiotic soap is a bad idea. Phototrophic bacteria, such as Chlorobium tepidum, that can convert H2S to H2SO4 exist in nature [Zerkle2009, Wahlund1991], for example in sulfur hot springs in Yellowstone Park. These highly specialized bacteria can convert the light energy from the sun into chemical energy in the sulfate ion.
Another possibility is that we have specialized cells in the skin, possibly the keratinocytes, that are able to exploit sunlight to convert sulfide to sulfate, using a similar phototrophic mechanism to C. tepidum. This seems quite plausible, especially considering that both human keratinocytes and C. tepidum can synthesize an interesting UV-B absorbing cofactor, tetrahydrobioptin. This cofactor is found universally in mammalian cells, and one of its roles is to regulate the synthesis of melanin [Schallreut94], the skin pigment that is associated with a tan and protects the skin from damage by UV-light exposure [Costin2007]. However, tetrahydrobiopsin is very rare in the bacterial kingdom, and C. tepidum is one of the very few bacteria that can synthesize it [Cho99].
Let me summarize at this point where I'm on solid ground and where I'm speculating. It is undisputed that the skin synthesizes cholesterol sulfate in large amounts, and it has been suggested that the skin is the major supplier of cholesterol sulfate to the blood stream [Strott2003]. The skin also synthesizes vitamin D3 sulfate, upon exposure to sunlight. Vitamin D3 is synthesized from cholesterol, with oxysterols (created from sun exposure) as an intermediate step (oxysterols are forms of cholesterol with hydroxyl groups attached at various places in the carbon chain). The body can't synthesize vitamin D3 sulfate from vitamin D3 [Lakdawala1977] so it must be that sulfation happens first, producing cholesterol sulfate or hydroxy-cholesterol sulfate, which is then optionally converted to vitamin D3 sulfate or shipped out "as is."
Another highly significant feature of skin cells is that the skin stores sulfate ions attached to molecules that are universally present in the intracellular matrix, such as heparan sulfate, chondroitin sulfate, and keratin sulfate [Milstone1994]. Furthermore, it has been shown that exposure of the melanin producing cells (melanocytes) to molecules containing reduced sulfur (-2) leads to suppression of melanin synthesis [Chu2009], whereas exposure to molecules like chondroitin sulfate that contain oxidized sulfur (+6) leads to enhancement of melanin synthesis [Katz1976]. Melanin is a potent UV-light absorber, and it would compete with reduced sulfur for the opportunity to become oxidized. It is therefore logical that, when sulfur is reduced, melanin synthesis should be suppressed, so that sulfur can absorb the solar energy and convert it to very useful chemical bonds in the sulfate ion.
The sulfate would eventually be converted back to sulfide by a muscle cell in the heart or a skeletal muscle (simultaneously recovering the energy to fuel the cell and unlocking the oxygen to support aerobic metabolism of glucose), and the cycle would continually repeat.
Why am I spending so much time talking about all of this? Well, if I'm right, then the skin can be viewed as a solar-powered battery for the heart, and that is a remarkable concept. The energy in sunlight is converted into chemical energy in the oxygen-sulfur bonds, and then transported through the blood vessels to the heart and skeletal muscles. The cholesterol sulfate and vitamin D3 sufate are carriers that deliver the energy (and the oxygen) "door-to-door" to the individual heart and skeletal muscle cells.
Today's lifestyle, especially in America, severely stresses this system. First of all, most Americans believe that any food containing cholesterol is unhealthy, so the diet is extremely low in cholesterol. Eggs are an excellent source of sulfur, but because of their high cholesterol content we have been advised to eat them sparingly. Secondly, as I discussed previously, natural food plant sources of sulfur are likely to be deficient due to sulfur depletion in the soil. Thirdly, water softeners remove sulfur from our water supply, which would otherwise be a good source. Fourthly, we have been discouraged from eating too much red meat, an excellent source of sulfur-containing amino acids. Finally, we have been instructed by doctors and other authoritarian sources to stay out of the sun and wear high SPF sunscreen whenever we do get sun exposure.
Another significant contributor is the high carbohydrate, low fat diet, which leads to excess glucose in the blood stream that glycates LDL particles and renders them ineffective in delivering cholesterol to the tissues. One of those tissues is the skin, so skin becomes further depleted in cholesterol due to glycation damage to LDL.
9. Sulfur Deficiency and Muscle Wasting Diseases
In browsing the Web, I recently came upon a remarkable article [Dröge1997] which develops a persuasive theory that low blood serum levels of two sulfur-containing molecules are a characteristic feature of a number of diseases/conditions. All of these diseases are associated with muscle wasting, despite adequate nutrition. The authors have coined the term "low CG syndrome" to represent this observed profile., where "CG" stands for the amino acid "cysteine," and the tripeptide "glutathione," both of which contain a sulfhydryl radical "-S-H" that is essential to their function. Glutathione is synthesized from the amino acids cysteine, glutamate, and glycine, and glutamate deficiency figures into the disease process as well, as I will discuss later.
The list of diseases/conditions associated with low CG syndrome is surprising and very revealing: HIV infection, cancer, major injuries, sepsis (blood poisoning), Crohn's disease (irritable bowel syndrome), ulcerative colitis, chronic fatigue syndrome, and athletic over-training. The paper [Drage1997] is dense but beautifully written, and it includes informative diagrams that explain the intricate feedback mechanisms between the liver and the muscles that lead to muscle wasting.
This paper fills in some missing holes in my theory, but the authors never suggest that sulfur deficiency might actually be a precursor to the development of low CG syndrome. I think that, particularly with respect to Crohn's disease, chronic fatigue syndrome, and excessive exercise, sulfur deficiency may precede and provoke the muscle wasting phenomenon. The biochemistry involved is complicated, but I will try to explain it in as simple terms as possible.
I will use Crohn's disease as my primary focus for discussion: an inflammation of the intestines, associated with a wide range of symptoms, including reduced appetite, low-grade fever, bowel inflammation, diarrhea, skin rashes, mouth sores, and swollen gums. Several of these symptoms suggest problems with the interface between the body and the external world: i.e., a vulnerability to invasive pathogens. I mentioned before that cholesterol sulfate plays a crucial role in the barrier that keeps pathogens from penetrating the skin. It logically plays a similar role everywhere there is an opportunity for bacteria to invade, and certainly a prime opportunity is available at the endothelial barrier in the intestines. Thus, I hypothesize that the intestinal inflammation and low-grade fever are due to an overactive immune system, necessitated by the fact that pathogens have easier access when the endothelial cells are deficient in cholesterol sulfate. The skin rashes and mouth and gum problems are a manifestation of inflammation elsewhere in the barrier.
Ordinarily, the liver supplies cholesterol sulfate to the gall bladder, where it is mixed into bile acids, and subsequently released into the digestive system to assist in the digestion of fats. If a person consistently eats a low-fat diet, the amount of cholesterol sulfate delivered to the digestive system from the liver will be reduced. This will logically result in a digestive system that is more vulnerable to invasion by pathogens.
The sulfate that's combined with cholesterol in the liver is synthesized from cysteine (one of the two proteins that are deficient in low CG syndome). So insufficient bioavailability of cysteine will lead to a reduced production of cholesterol sulfate by the liver. This will, in turn, make it difficult to digest fats, likely, over time, compelling the person to adhere to a low-fat diet. Whether low-fat diet or sulfur deficiency comes first, the end result is a vulnerability to infective agents in the intestines, with a consequential heightened immune response.
[Dröge1997] further discussses how a reduction in the synthesis of sulfate from cysteine in the liver leads to increased compensatory activity in another biological pathway in the liver that converts glutamate to arginine and urea. Glutamate is highly significant because it is produced mainly by the breakdown of amino acids (proteins in the muscles); i.e., by muscle wasting. The muscle cells are triggered to cannibalize themselves in order to provide adequate glutamate to the liver, mainly, in my view, in order to generate enough arginine to replace the role of sulfate in muscle glucose metabolism (i.e., these activities in the liver and muscles are circular and mutually supportive).
Arginine is the major source of nitric oxide (NO) and NO is the next best thing for muscle glucose metabolism in the absence of cholesterol sulfate. NO is a poor substitue for SO4-2, but it can function in some of the missing roles. As you will recall, I propose that cholesterol SO4-2 accomplishes a number of important things in muscle cells: it delivers oxygen to myoglobin, it supplies cholesterol to the cell membrane, it helps break down glucose, protects the cell's proteins from glycation and oxidation damage, and provides energy to the cell. NO can help in reducing glycation damage, as nitrogen can be reduced from +2 to 0 (whereas sulfur was reduced from +6 to -2). It also provides oxygen, but it is unable to transfer the oxygen directly to myoglobin by binding with the iron molecule, as was the case for sulfate. NO does not supply cholesterol, so cholesterol deficiency remains a problem, leaving the cell's proteins and fats more vulnerable to oxidative damage. Furthermore, NO itself is an oxidizing agent, so myoglobin becomes disabled, due to both oxidation and glycation damage. The muscle cell, therefore, engages in mitochondrial oxidation of glucose at its own peril: better to revert to anaerobic metabolism of glucose to decrease the risk of damage. Anaerobic metabolism of glucose results in a build-up of lactic acid, which, as explained in [Dröge1997] further enhances the need for the liver to metabolize glutamate, thus augmenting the feedback loop.
Furthermore, as you'll recall, if I'm right about cholesterol sulfate seeding lipid rafts, then, with a cholesterol sulfate deficiency, the entry of both glucose and fat into the muscle cell are compromised. This situation leaves the cell with little choice but to exploit its internal proteins as fuel, manifested as muscle wasting.
In summary, a number of different arguments lead to the hypothesis that sulfur deficiency causes the liver to shift from producing cholesterol sulfate to producing arginine (and subsequently nitric oxide). This leaves the intestines and muscle cells vulnerable to oxidation damage, which can explain both the intestinal inflammation and the muscle wasting associated with Crohn's disease.
The immune system depends upon abundant cholesterol to defend against severe stress. I have previously argued that high serum cholesterol is protective against sepsis. It is worth repeating here the abstract from [Wilson2003], who studied changes in blood cholesterol levels following trauma, infection, and multiple organ failure:
"Hypocholesterolemia is an important observation following trauma. In a study of critically ill trauma patients, mean cholesterol levels were significantly lower (119 ± 44 mg/dl) than expected values (201 ± 17 mg/dl). In patients who died, final cholesterol levels fell by 33% versus a 28% increase in survivors. Cholesterol levels were also adversely affected by infection or organ system dysfunction. Other studies have illustrated the clinical significance of hypocholesterolemia. Because lipoproteins can bind and neutralize lipopolysaccharide, hypocholesterolemia can negatively impact outcome. New therapies directed at increasing low cholesterol levels may become important options for the treatment of sepsis."
Thus, many of these conditions/diseases that lead to muscle wasting may do so because cholesterol (and therefore cholesterol sulfate) is depleted from the blood serum. This results in the same feedback loop between the liver and the muscles that I discussed with regard to Crohn's disease. So I think it's plausible that the muscle wasting associated with all of these conditions is caused by this same feedback mechanism.
I have discussed the role cysteine plays in providing sulfate to the liver. But what is the role of glutathione, the other sulfur-containing protein that's depleted in low GC syndrome? Muscle cells ordinarily contain significant levels of glutathione, and its depletion leads to mitochondrial damage [Martensson1989]. Patients undergoing surgical trauma have been found to exhibit reduced glutathione levels in their skeletal muscles [Luo1996]. It is tempting to speculate that cholesterol sulfate provides the sulfur needed for glutathione synthesis, so that the deficiency would be explained by the reduced availability of cholesterol following the immune system's heightened response to surgical trauma. Glutathione is a potent antioxidant, so its deficiency will further contribute to dysfunction of the muscle cell's mitochondria, therefore greatly impairing its energy supply.
There is a growing awareness that glutathione deficiency may play a role in many diseases. You may want to check out this Web site describing a long list of diseases that may be impacted by glutathione deficiency. Whether the problems arise just due to insufficient supply of the glutathione molecule itself, or whether a more general sulfur deficiency is the root cause, is perhaps hard to say, but provocative nonetheless.
The list of diseases/conditions associated with low CG syndrome is surprising and very revealing: HIV infection, cancer, major injuries, sepsis (blood poisoning), Crohn's disease (irritable bowel syndrome), ulcerative colitis, chronic fatigue syndrome, and athletic over-training. The paper [Drage1997] is dense but beautifully written, and it includes informative diagrams that explain the intricate feedback mechanisms between the liver and the muscles that lead to muscle wasting.
This paper fills in some missing holes in my theory, but the authors never suggest that sulfur deficiency might actually be a precursor to the development of low CG syndrome. I think that, particularly with respect to Crohn's disease, chronic fatigue syndrome, and excessive exercise, sulfur deficiency may precede and provoke the muscle wasting phenomenon. The biochemistry involved is complicated, but I will try to explain it in as simple terms as possible.
I will use Crohn's disease as my primary focus for discussion: an inflammation of the intestines, associated with a wide range of symptoms, including reduced appetite, low-grade fever, bowel inflammation, diarrhea, skin rashes, mouth sores, and swollen gums. Several of these symptoms suggest problems with the interface between the body and the external world: i.e., a vulnerability to invasive pathogens. I mentioned before that cholesterol sulfate plays a crucial role in the barrier that keeps pathogens from penetrating the skin. It logically plays a similar role everywhere there is an opportunity for bacteria to invade, and certainly a prime opportunity is available at the endothelial barrier in the intestines. Thus, I hypothesize that the intestinal inflammation and low-grade fever are due to an overactive immune system, necessitated by the fact that pathogens have easier access when the endothelial cells are deficient in cholesterol sulfate. The skin rashes and mouth and gum problems are a manifestation of inflammation elsewhere in the barrier.
Ordinarily, the liver supplies cholesterol sulfate to the gall bladder, where it is mixed into bile acids, and subsequently released into the digestive system to assist in the digestion of fats. If a person consistently eats a low-fat diet, the amount of cholesterol sulfate delivered to the digestive system from the liver will be reduced. This will logically result in a digestive system that is more vulnerable to invasion by pathogens.
The sulfate that's combined with cholesterol in the liver is synthesized from cysteine (one of the two proteins that are deficient in low CG syndome). So insufficient bioavailability of cysteine will lead to a reduced production of cholesterol sulfate by the liver. This will, in turn, make it difficult to digest fats, likely, over time, compelling the person to adhere to a low-fat diet. Whether low-fat diet or sulfur deficiency comes first, the end result is a vulnerability to infective agents in the intestines, with a consequential heightened immune response.
[Dröge1997] further discussses how a reduction in the synthesis of sulfate from cysteine in the liver leads to increased compensatory activity in another biological pathway in the liver that converts glutamate to arginine and urea. Glutamate is highly significant because it is produced mainly by the breakdown of amino acids (proteins in the muscles); i.e., by muscle wasting. The muscle cells are triggered to cannibalize themselves in order to provide adequate glutamate to the liver, mainly, in my view, in order to generate enough arginine to replace the role of sulfate in muscle glucose metabolism (i.e., these activities in the liver and muscles are circular and mutually supportive).
Arginine is the major source of nitric oxide (NO) and NO is the next best thing for muscle glucose metabolism in the absence of cholesterol sulfate. NO is a poor substitue for SO4-2, but it can function in some of the missing roles. As you will recall, I propose that cholesterol SO4-2 accomplishes a number of important things in muscle cells: it delivers oxygen to myoglobin, it supplies cholesterol to the cell membrane, it helps break down glucose, protects the cell's proteins from glycation and oxidation damage, and provides energy to the cell. NO can help in reducing glycation damage, as nitrogen can be reduced from +2 to 0 (whereas sulfur was reduced from +6 to -2). It also provides oxygen, but it is unable to transfer the oxygen directly to myoglobin by binding with the iron molecule, as was the case for sulfate. NO does not supply cholesterol, so cholesterol deficiency remains a problem, leaving the cell's proteins and fats more vulnerable to oxidative damage. Furthermore, NO itself is an oxidizing agent, so myoglobin becomes disabled, due to both oxidation and glycation damage. The muscle cell, therefore, engages in mitochondrial oxidation of glucose at its own peril: better to revert to anaerobic metabolism of glucose to decrease the risk of damage. Anaerobic metabolism of glucose results in a build-up of lactic acid, which, as explained in [Dröge1997] further enhances the need for the liver to metabolize glutamate, thus augmenting the feedback loop.
Furthermore, as you'll recall, if I'm right about cholesterol sulfate seeding lipid rafts, then, with a cholesterol sulfate deficiency, the entry of both glucose and fat into the muscle cell are compromised. This situation leaves the cell with little choice but to exploit its internal proteins as fuel, manifested as muscle wasting.
In summary, a number of different arguments lead to the hypothesis that sulfur deficiency causes the liver to shift from producing cholesterol sulfate to producing arginine (and subsequently nitric oxide). This leaves the intestines and muscle cells vulnerable to oxidation damage, which can explain both the intestinal inflammation and the muscle wasting associated with Crohn's disease.
The immune system depends upon abundant cholesterol to defend against severe stress. I have previously argued that high serum cholesterol is protective against sepsis. It is worth repeating here the abstract from [Wilson2003], who studied changes in blood cholesterol levels following trauma, infection, and multiple organ failure:
"Hypocholesterolemia is an important observation following trauma. In a study of critically ill trauma patients, mean cholesterol levels were significantly lower (119 ± 44 mg/dl) than expected values (201 ± 17 mg/dl). In patients who died, final cholesterol levels fell by 33% versus a 28% increase in survivors. Cholesterol levels were also adversely affected by infection or organ system dysfunction. Other studies have illustrated the clinical significance of hypocholesterolemia. Because lipoproteins can bind and neutralize lipopolysaccharide, hypocholesterolemia can negatively impact outcome. New therapies directed at increasing low cholesterol levels may become important options for the treatment of sepsis."
Thus, many of these conditions/diseases that lead to muscle wasting may do so because cholesterol (and therefore cholesterol sulfate) is depleted from the blood serum. This results in the same feedback loop between the liver and the muscles that I discussed with regard to Crohn's disease. So I think it's plausible that the muscle wasting associated with all of these conditions is caused by this same feedback mechanism.
I have discussed the role cysteine plays in providing sulfate to the liver. But what is the role of glutathione, the other sulfur-containing protein that's depleted in low GC syndrome? Muscle cells ordinarily contain significant levels of glutathione, and its depletion leads to mitochondrial damage [Martensson1989]. Patients undergoing surgical trauma have been found to exhibit reduced glutathione levels in their skeletal muscles [Luo1996]. It is tempting to speculate that cholesterol sulfate provides the sulfur needed for glutathione synthesis, so that the deficiency would be explained by the reduced availability of cholesterol following the immune system's heightened response to surgical trauma. Glutathione is a potent antioxidant, so its deficiency will further contribute to dysfunction of the muscle cell's mitochondria, therefore greatly impairing its energy supply.
There is a growing awareness that glutathione deficiency may play a role in many diseases. You may want to check out this Web site describing a long list of diseases that may be impacted by glutathione deficiency. Whether the problems arise just due to insufficient supply of the glutathione molecule itself, or whether a more general sulfur deficiency is the root cause, is perhaps hard to say, but provocative nonetheless.
9. Sulfur Deficiency and Muscle Wasting Diseases
A serious disorder known as ("Smith-Lemli-Opitz") syndrome (SLOS) is characterized by a genetic defect resulting in an inability to synthesize adequate cholesterol. Most fetuses that are unfortunate to be conceived with this genetic defect don't make it to 16 weeks of gestation before the pregnancy ends in a miscarriage. If they do manage to make it to term, they typically suffer from major brain defects, resulting in autism or other forms of mental retardation [9].
10. Summary
Although sulfur is an essential element in human biology, we hear surprisingly little about sulfur in discussions on health. Sulfur binds strongly with oxygen, and is able to stably carry a charge ranging from +6 to -2, and is therefore very versatile in supporting aerobic metabolism. There is strong evidence that sulfur deficiency plays a role in diseases ranging from Alzheimer's to cancer to heart disease. Particularly intriguing is the relationship between sulfur deficiency and muscle wasting, a signature of end-stage cancer, AIDS, Crohn's disease, and chronic fatigue syndrome.
The African rift zone, where humans are believed to have first made their appearance several million years ago, would have been rich with sulfur supplied by active volcanism. It is striking that people living today in places where sulfur is abundantly provided by recent volcanism enjoy a low risk for heart disease and obesity.
In my research on sulfur, I was drawn to two mysterious molecules: cholesterol sulfate and vitamin D3 sulfate. Researchers have not yet determined the role that cholesterol sulfate plays in the blood stream, despite the fact that it is ubiquitous there. Research experiments have clearly shown that cholesterol sulfate is protective against heart disease. I have developed a theory proposing that cholesterol sulfate is central to the formation of lipid rafts, which, in turn, are essential for aerobic glucose metabolism. I would predict that deficiencies in cholesterol sulfate lead to severe defects in muscle metabolism, and this includes the heart muscle. My theory would explain the protective role of cholesterol sulfate in heart disease and muscle wasting diseases.
I have also argued that cholesterol sulfate delivers oxygen to myoglobin in muscle cells, resulting in safe oxygen transport to the mitochondria. I argue a similar role for alpha-synuclein in the brain. There is a striking relationship between Alzheimer's and sulfur depletion in neurons in the brain. Sulfur plays a key role in protectiing proteins in neurons and muscle cells from oxidative damage, while maintaining adequate oxygen supply to the mitochondria.
When muscles become impaired in glucose metabolism due to reduced availability of cholesterol sulfate, proliferating fat cells become involved in converting glucose to fat. This provides an alternative fuel for the muscle cells, and replenishes the cholesterol supply by storing and refurbishing cholesterol extracted from defective LDL. Thin people with cholesterol and sulfur deficiency are vulnerable to a wide range of problems, such as Crohn's disease, chronic fatigue syndrome, and muscle wasting, because fat cells are not available to ameliorate the situation.
Cholesterol sulfate in the epithelium protects from invasion of pathogens through the skin, which greatly reduces the burden placed on the immune system. Perhaps the most intriguing possibility presented here is the idea that sulfur provides a way for the skin to become a solar-powered battery: to store the energy from sunlight as chemical energy in the sulfate molecule. This seems like a very sensible and practical scheme, and the biochemistry involved has been demonstrated to work in phototrophic sulfur-metabolizing bacteria found in sulfur hot springs.
The skin produces vitamin D3 sulfate upon exposure to sunlight, and the vitamin D3 found in breast milk is also sulfated. In light of these facts, it is quite surprising to me that so little research has been directed towards understanding what role sulfated vitamin D3 plays in the body. It is recently becoming apparent that vitamin D3 promotes a strong immune system and offers protection against cancer, yet how it achieves these benefits is not at all clear. I strongly suspect that it is vitamin D3 sulfate that carries out this aspect of vitamin D3's positive influence.
Modern lifestyle practices conspire to induce major deficiencies in cholesterol sulfate and vitamin D3 sulfate. We are encouraged to actively avoid sun exposure and to minimize dietary intake of cholesterol-containing foods. We are encouraged to consume a high-carbohydrate/low-fat diet which, as I have argued previously (Seneff2010), leads to impaired cholesterol uptake in cells. We are told nothing about sulfur, yet many factors, ranging from the Clean Air Act to intensive farming to water softeners, deplete the supply of sulfur in our food and water.
Fortunately, correcting these deficiencies at the individual level is easy and straightforward. If you just throw away the sunscreen and eat more eggs, those two steps alone may greatly increase your chances of living a long and healthy life.

Could Sulfur Deficiency be a Contributing Factor to Obesity, Heart Disease, Alzheimer's and Chronic Fatigue Syndrome? by Stephanie Seneff is licensed under a Creative Commons Attribution 3.0 United States License.
The African rift zone, where humans are believed to have first made their appearance several million years ago, would have been rich with sulfur supplied by active volcanism. It is striking that people living today in places where sulfur is abundantly provided by recent volcanism enjoy a low risk for heart disease and obesity.
In my research on sulfur, I was drawn to two mysterious molecules: cholesterol sulfate and vitamin D3 sulfate. Researchers have not yet determined the role that cholesterol sulfate plays in the blood stream, despite the fact that it is ubiquitous there. Research experiments have clearly shown that cholesterol sulfate is protective against heart disease. I have developed a theory proposing that cholesterol sulfate is central to the formation of lipid rafts, which, in turn, are essential for aerobic glucose metabolism. I would predict that deficiencies in cholesterol sulfate lead to severe defects in muscle metabolism, and this includes the heart muscle. My theory would explain the protective role of cholesterol sulfate in heart disease and muscle wasting diseases.
I have also argued that cholesterol sulfate delivers oxygen to myoglobin in muscle cells, resulting in safe oxygen transport to the mitochondria. I argue a similar role for alpha-synuclein in the brain. There is a striking relationship between Alzheimer's and sulfur depletion in neurons in the brain. Sulfur plays a key role in protectiing proteins in neurons and muscle cells from oxidative damage, while maintaining adequate oxygen supply to the mitochondria.
When muscles become impaired in glucose metabolism due to reduced availability of cholesterol sulfate, proliferating fat cells become involved in converting glucose to fat. This provides an alternative fuel for the muscle cells, and replenishes the cholesterol supply by storing and refurbishing cholesterol extracted from defective LDL. Thin people with cholesterol and sulfur deficiency are vulnerable to a wide range of problems, such as Crohn's disease, chronic fatigue syndrome, and muscle wasting, because fat cells are not available to ameliorate the situation.
Cholesterol sulfate in the epithelium protects from invasion of pathogens through the skin, which greatly reduces the burden placed on the immune system. Perhaps the most intriguing possibility presented here is the idea that sulfur provides a way for the skin to become a solar-powered battery: to store the energy from sunlight as chemical energy in the sulfate molecule. This seems like a very sensible and practical scheme, and the biochemistry involved has been demonstrated to work in phototrophic sulfur-metabolizing bacteria found in sulfur hot springs.
The skin produces vitamin D3 sulfate upon exposure to sunlight, and the vitamin D3 found in breast milk is also sulfated. In light of these facts, it is quite surprising to me that so little research has been directed towards understanding what role sulfated vitamin D3 plays in the body. It is recently becoming apparent that vitamin D3 promotes a strong immune system and offers protection against cancer, yet how it achieves these benefits is not at all clear. I strongly suspect that it is vitamin D3 sulfate that carries out this aspect of vitamin D3's positive influence.
Modern lifestyle practices conspire to induce major deficiencies in cholesterol sulfate and vitamin D3 sulfate. We are encouraged to actively avoid sun exposure and to minimize dietary intake of cholesterol-containing foods. We are encouraged to consume a high-carbohydrate/low-fat diet which, as I have argued previously (Seneff2010), leads to impaired cholesterol uptake in cells. We are told nothing about sulfur, yet many factors, ranging from the Clean Air Act to intensive farming to water softeners, deplete the supply of sulfur in our food and water.
Fortunately, correcting these deficiencies at the individual level is easy and straightforward. If you just throw away the sunscreen and eat more eggs, those two steps alone may greatly increase your chances of living a long and healthy life.

Could Sulfur Deficiency be a Contributing Factor to Obesity, Heart Disease, Alzheimer's and Chronic Fatigue Syndrome? by Stephanie Seneff is licensed under a Creative Commons Attribution 3.0 United States License.
References for Sulfur Essay
1. Axelson1985
Magnus Axelson, "25-Hydroxyvitamin D3 3-sulphate is a major circulating form of vitamin D in man," FEBS Letters (1985), Volume 191, Issue 2, 28 October, Pages 171-175; doi:10.1016/0014-5793(85)80002-8
2. Crawford1967
T. Crawford and Margaret D. Crawford, "Prevalence and Pathological Changes of Ischaemic Heart-Disease in a Hard-water and in a Soft-water Area," The Lancet (1967) Saturday 4 February
3. Biorck1965
Biorck, G., Bostrom, H., Widstrom, A. "Trace Elements and Cardiovascular Diseases", Acta med. scand. (1965) 178, 239.
4. Brownlee1988
Brownlee M, Cerami A and Vlassara H. "Advanced glycosylation end products in tissue and the biochemical basis of diabetic complications." N Engl J Med (1988) 318: pp. 1315¬1321.
5. Brown1936
"W. R. Brown, the hydrolysis of starch by hydrogen peroxide and ferrous sulfate." J. Biol. Chem. (1936) 113: 417-425.
6. Boulch1982
N Le Boulch, L. Cancela and L. Miravet, "Cholecalciferol sulfate identification in human milk by HPLC," Steroids (1982) Volume 39, Issue 4, April, Pages 391-398; doi:10.1016/0039-128X(82)90063-0
7. Cho99
Cho SH, Na JU, Youn H, Hwang CS, Lee CH, Kang SO, "Sepiapterin reductase producing L-threo-dihydrobiopterin from Chlorobium tepidum." Biochem J (1999) 340 ( Pt g2);497-503. PMID: 10333495
8. Cinti2005
Cinti S, Mitchell G, Barbatelli G, Murano I, Ceresi E, Faloia E, Wang S, Fortier M, Greenberg AS and Obin MS. "Adipocyte death defines macrophage localization and function in adipose tissue of obese mice and humans." J Lipid Res (2005) 46: pp. 2347-2355.
9. Costin2007
Gertrude-E. Costin and Vincent J. Hearing, "Human skin pigmentation: melanocytes modulate skin color in response to stress," The FASEB Journal (2007), 21:976-994; doi: 10.1096/fj.06-6649rev.
10. Chu2009
Heuy-Ling Chu, Bor-Sen Wang and Pin-Der Duh, "Effects of Selected Organo-sulfur Compounds on Melanin Formation," J. Agric. Food Chem. (2009) 57 (15), pp 7072–7077; DOI: 10.1021/jf9005824.
11. Dröge1997
Wulf Dröge and Eggert Holm, "Role of cysteine and glutathione in H1V infection and other diseases associated with muscle wasting and immunological dysfunction," The FASEB Journal (1997) Vol. 11, November, pp. 1077-1089.
12. Drolet2003
Marie-Claude Drolet, Marie Arsenault, and Jacques Couet, "Experimental Aortic Valve Stenosis in Rabbits," J. Am. Coll. Cardiol. (2003) Vol. 41, pp. 1211-1217.
13. Glaser2005
Charles B. Glaser, Ghiam Yamin, Vladimir N. Uversky, and Anthony L. Fink, "Methionine oxidation, a-synuclein and Parkinson’s disease," Biochimica et Biophysica Acta (2005) Vol. 1703, pp. 157–169
14. Grimes1996
D.S. Grimes, E. Hindle, and T. Dyer, "Sunlight, cholesterol and coronary heart disease." Q. J. Med. (1996) 89:579-589.
15. Hockin2003
Simon L. Hockin and Geoffrey M. Gadd, "Linked Redox Precipitation of Sulfur and Selenium under Anaerobic Conditions by Sulfate-Reducing Bacterial Biofilms," Applied and Environmental Microbiology (2003) Dec., p. 7063–7072, Vol. 69, No. 12; DOI: 10.1128/AEM.69.12.7063–7072.2003
16. Inoue2006
Inoue, M., Chiang, S.H., Chang, L., Chen, X.W. and Saltiel, A.R. "Compartmentalization of the exocyst complex in lipid rafts controls Glut4 vesicle tethering." Mol. Biol. Cell (2006) 17, 2303–2311
17. Jez2008
Joseph Jez, "Sulfur: a Missing Link between Soils, Crops, and Nutrition." Agronomy Monograph #50. (2008) American Society of Agronomy, Inc. Crop Science Society of America, Inc., Soil Science Society of American, Inc.
18.Katz1976
Katz IR, Yamauchi T, Kaufman S. "Activation of tyrosine hydroxylase by polyanions and salts. An electrostatic effect." Biochim Biophys Acta. (1976) Mar 11;429(1):84-95.
19.Lakdawala1977
Dilnawaz R. Lakdawala and Elsie M. Widdowson, "Vitamin D in Human Milk," The Lancet (1977) Volume 309, Issue 8004, 22 January, Pages 167-168.
20.Li1997
Yong Ming Li and Dennis W. Dickson, "Enhanced binding of advanced glycation endproducts (AGE) by the ApoE4 isoform links the mechanism of plaque deposition in Alzheimer's disease," Neuroscience Letters (1997), Volume 226, Issue 3, 2 May, Pages 155-158; doi:10.1016/S0304-3940(97)00266-8
21.Luo1996
J L Luo, F Hammarqvist, K Andersson, and J Wernerman, "Skeletal muscle glutathione after surgical trauma." Ann Surg. (1996) April; 223(4): 420–427.
22.Ma2008
Yongjie Ma, Leyuan Xu, Daniel Rodriguez-Agudo, Xiaobo Li, Douglas M. Heuman, Phillip B. Hylemon, William M. Pandak and Shunlin Ren, "25-Hydroxycholesterol-3-sulfate regulates macrophage lipid metabolism via the LXR/SREBP-1 signaling pathway," Am J Physiol Endocrinol Metab (2008) 295:1369-1379; doi:10.1152/ajpendo.90555.2008
23.Martensson1989
Martensson, J., and Meister,A., "Mitochondrial damage in muscle occurs after marked depletion of glutathione and is prevented by giving glutathione monoester." Proc Natl Acad Sci U S A, (1989) 86:471-475.
24.McGrath2008
John A. McGrath and Jouni Uitto "The filaggrin story: novel insights into skin-barrier function and disease," Trends in Molecular Medicine (2008) Volume 14, Issue 1, January, Pages 20-27.
25.Milstone1994
Leonard M. Milstone, Lynne Hough-Monroe, Lisa C. Kugelman, Jeffrey R. Bender and John G. Haggerty, "Epican, a heparan/chondroitin sulfate proteoglycan form of CD44, mediates cell-cell adhesion," Journal of Cell Science (1994) 107, 3183-3190
26. Ojuka2002
E.O. Ojuka, T.E. Jones, L.A. Nolte, M. Chen, B.R. Wamhoff, M. Sturek, and J.O. Holloszy, "Regulation of GLUT4 biogenesis in muscle: evidence for involvement of AMPK and Ca2+," Am J Physiol Endocrinol Metab (2002) Vol. 282, NO. 5, May.
27. Olivares2009
Olivares D, Huang X, Branden L, Greig NH, Rogers JT. "Physiological and Pathological Role of Alpha-synuclein in Parkinson's Disease Through Iron Mediated Oxidative Stress; The Role of a Putative Iron-responsive Element," Int J Mol Sci (2009) 10:1226-60.
28. Reeve1981
Lorraine E. Reeve, Hector F. DeLuca, and Heinrich K. Schnoes, "Synthesis and Biological Activity of Vitamin D3-Sulfate," The Journal of Biological Chemistry (1981) Vol. 256., NO. 2. Jan 25, pp. 823-826.
29. Rodriguez1995
W. V. Rodriguez, J. J. Wheeler, S. K. I.imuk, C. N. Kitson, and M. J. Hope, "Transbilayer Movement and Net Flux of Cholesterol and Cholesterol Sulfate between Liposomal Membranes", Biochemistry (1995) 34, 6208-6217.
30. Sandilands2009
Sandilands A, Sutherland C, Irvine AD, McLean WH, "Filaggrin in the frontline: role in skin barrier function and disease," J Cell Sci. (2009) May 1;122(Pt 9):1285-94.
31. Scappola1995
Scoppola A, Testa G, Frontoni S, Maddaloni E, Gambardella S, Menzinger G and Lala A. "Effects of insulin on cholesterol synthesis in type II diabetes patients," Diabetes Care (1995) 18: pp. 1362-1369.
32. Schallreut94
Schallreuter KU, Wood JM, Pittelkow MR, Gutlich M, Lemke KR, Rodl W, Swanson NN, Hitzemann K, Ziegler I, "Regulation of melanin biosynthesis in the human epidermis by tetrahydrobiopterin." Science (1994) 263(5152);1444-6. PMID: 8128228
33. Seneff2010
S. Seneff, G. Wainwright, and L. Mascitelli, "Is the metabolic syndrome caused by a high fructose, and relatively low fat, low cholesterol diet?", Archives of Medical Science (2010), To Appear.
34. Strott2003
Charles A. Strott and Yuko Higashi, "Cholesterol sulfate in human physiology: what's it all about?" Journal of Lipid Research (2003) Volume 44, pp. 1268-1278.
35. Wahlund1991
Wahlund, T. M., C. R. Woese, R. W. Castenholz, and M. T. Madigan, "A thermophilic green sulfur bacterium from New Zealand hot springs, Chlorobium tepidum sp." Nov. Arch. Microbiol. (1991) 159:81-90.
36. Waldman2009
M. Waldman, MD,, 9th International Conference on Alzheimer's and Parkinson's Diseases (2009) Abstract 90, Presented March 12-13.
37. Wilson2003
Robert F Wilson, Jeffrey F Barletta and James G Tyburski,"Hypocholesterolemia in Sepsis and Critically Ill or Injured Patients" Critical Care 7:413-414, 2003. http://www.medscape.com/viewarticle/511735_2
38. Zerkle2009
Aubrey L. Zerkle, James Farquhar, David T. Johnston, Raymond P. Cox, and Donald E. Canfield, "Fractionation of multiple sulfur isotopes during phototrophic oxidation of sulfide and elemental sulfur by a green sulfur bacterium," Geochimica et Cosmochimica Acta (2009) Volume 73, Issue 2, 15 January 2009, Pages 291-306; doi:10.1016/j.gca.2008.10.027
Magnus Axelson, "25-Hydroxyvitamin D3 3-sulphate is a major circulating form of vitamin D in man," FEBS Letters (1985), Volume 191, Issue 2, 28 October, Pages 171-175; doi:10.1016/0014-5793(85)80002-8
2. Crawford1967
T. Crawford and Margaret D. Crawford, "Prevalence and Pathological Changes of Ischaemic Heart-Disease in a Hard-water and in a Soft-water Area," The Lancet (1967) Saturday 4 February
3. Biorck1965
Biorck, G., Bostrom, H., Widstrom, A. "Trace Elements and Cardiovascular Diseases", Acta med. scand. (1965) 178, 239.
4. Brownlee1988
Brownlee M, Cerami A and Vlassara H. "Advanced glycosylation end products in tissue and the biochemical basis of diabetic complications." N Engl J Med (1988) 318: pp. 1315¬1321.
5. Brown1936
"W. R. Brown, the hydrolysis of starch by hydrogen peroxide and ferrous sulfate." J. Biol. Chem. (1936) 113: 417-425.
6. Boulch1982
N Le Boulch, L. Cancela and L. Miravet, "Cholecalciferol sulfate identification in human milk by HPLC," Steroids (1982) Volume 39, Issue 4, April, Pages 391-398; doi:10.1016/0039-128X(82)90063-0
7. Cho99
Cho SH, Na JU, Youn H, Hwang CS, Lee CH, Kang SO, "Sepiapterin reductase producing L-threo-dihydrobiopterin from Chlorobium tepidum." Biochem J (1999) 340 ( Pt g2);497-503. PMID: 10333495
8. Cinti2005
Cinti S, Mitchell G, Barbatelli G, Murano I, Ceresi E, Faloia E, Wang S, Fortier M, Greenberg AS and Obin MS. "Adipocyte death defines macrophage localization and function in adipose tissue of obese mice and humans." J Lipid Res (2005) 46: pp. 2347-2355.
9. Costin2007
Gertrude-E. Costin and Vincent J. Hearing, "Human skin pigmentation: melanocytes modulate skin color in response to stress," The FASEB Journal (2007), 21:976-994; doi: 10.1096/fj.06-6649rev.
10. Chu2009
Heuy-Ling Chu, Bor-Sen Wang and Pin-Der Duh, "Effects of Selected Organo-sulfur Compounds on Melanin Formation," J. Agric. Food Chem. (2009) 57 (15), pp 7072–7077; DOI: 10.1021/jf9005824.
11. Dröge1997
Wulf Dröge and Eggert Holm, "Role of cysteine and glutathione in H1V infection and other diseases associated with muscle wasting and immunological dysfunction," The FASEB Journal (1997) Vol. 11, November, pp. 1077-1089.
12. Drolet2003
Marie-Claude Drolet, Marie Arsenault, and Jacques Couet, "Experimental Aortic Valve Stenosis in Rabbits," J. Am. Coll. Cardiol. (2003) Vol. 41, pp. 1211-1217.
13. Glaser2005
Charles B. Glaser, Ghiam Yamin, Vladimir N. Uversky, and Anthony L. Fink, "Methionine oxidation, a-synuclein and Parkinson’s disease," Biochimica et Biophysica Acta (2005) Vol. 1703, pp. 157–169
14. Grimes1996
D.S. Grimes, E. Hindle, and T. Dyer, "Sunlight, cholesterol and coronary heart disease." Q. J. Med. (1996) 89:579-589.
15. Hockin2003
Simon L. Hockin and Geoffrey M. Gadd, "Linked Redox Precipitation of Sulfur and Selenium under Anaerobic Conditions by Sulfate-Reducing Bacterial Biofilms," Applied and Environmental Microbiology (2003) Dec., p. 7063–7072, Vol. 69, No. 12; DOI: 10.1128/AEM.69.12.7063–7072.2003
16. Inoue2006
Inoue, M., Chiang, S.H., Chang, L., Chen, X.W. and Saltiel, A.R. "Compartmentalization of the exocyst complex in lipid rafts controls Glut4 vesicle tethering." Mol. Biol. Cell (2006) 17, 2303–2311
17. Jez2008
Joseph Jez, "Sulfur: a Missing Link between Soils, Crops, and Nutrition." Agronomy Monograph #50. (2008) American Society of Agronomy, Inc. Crop Science Society of America, Inc., Soil Science Society of American, Inc.
18.Katz1976
Katz IR, Yamauchi T, Kaufman S. "Activation of tyrosine hydroxylase by polyanions and salts. An electrostatic effect." Biochim Biophys Acta. (1976) Mar 11;429(1):84-95.
19.Lakdawala1977
Dilnawaz R. Lakdawala and Elsie M. Widdowson, "Vitamin D in Human Milk," The Lancet (1977) Volume 309, Issue 8004, 22 January, Pages 167-168.
20.Li1997
Yong Ming Li and Dennis W. Dickson, "Enhanced binding of advanced glycation endproducts (AGE) by the ApoE4 isoform links the mechanism of plaque deposition in Alzheimer's disease," Neuroscience Letters (1997), Volume 226, Issue 3, 2 May, Pages 155-158; doi:10.1016/S0304-3940(97)00266-8
21.Luo1996
J L Luo, F Hammarqvist, K Andersson, and J Wernerman, "Skeletal muscle glutathione after surgical trauma." Ann Surg. (1996) April; 223(4): 420–427.
22.Ma2008
Yongjie Ma, Leyuan Xu, Daniel Rodriguez-Agudo, Xiaobo Li, Douglas M. Heuman, Phillip B. Hylemon, William M. Pandak and Shunlin Ren, "25-Hydroxycholesterol-3-sulfate regulates macrophage lipid metabolism via the LXR/SREBP-1 signaling pathway," Am J Physiol Endocrinol Metab (2008) 295:1369-1379; doi:10.1152/ajpendo.90555.2008
23.Martensson1989
Martensson, J., and Meister,A., "Mitochondrial damage in muscle occurs after marked depletion of glutathione and is prevented by giving glutathione monoester." Proc Natl Acad Sci U S A, (1989) 86:471-475.
24.McGrath2008
John A. McGrath and Jouni Uitto "The filaggrin story: novel insights into skin-barrier function and disease," Trends in Molecular Medicine (2008) Volume 14, Issue 1, January, Pages 20-27.
25.Milstone1994
Leonard M. Milstone, Lynne Hough-Monroe, Lisa C. Kugelman, Jeffrey R. Bender and John G. Haggerty, "Epican, a heparan/chondroitin sulfate proteoglycan form of CD44, mediates cell-cell adhesion," Journal of Cell Science (1994) 107, 3183-3190
26. Ojuka2002
E.O. Ojuka, T.E. Jones, L.A. Nolte, M. Chen, B.R. Wamhoff, M. Sturek, and J.O. Holloszy, "Regulation of GLUT4 biogenesis in muscle: evidence for involvement of AMPK and Ca2+," Am J Physiol Endocrinol Metab (2002) Vol. 282, NO. 5, May.
27. Olivares2009
Olivares D, Huang X, Branden L, Greig NH, Rogers JT. "Physiological and Pathological Role of Alpha-synuclein in Parkinson's Disease Through Iron Mediated Oxidative Stress; The Role of a Putative Iron-responsive Element," Int J Mol Sci (2009) 10:1226-60.
28. Reeve1981
Lorraine E. Reeve, Hector F. DeLuca, and Heinrich K. Schnoes, "Synthesis and Biological Activity of Vitamin D3-Sulfate," The Journal of Biological Chemistry (1981) Vol. 256., NO. 2. Jan 25, pp. 823-826.
29. Rodriguez1995
W. V. Rodriguez, J. J. Wheeler, S. K. I.imuk, C. N. Kitson, and M. J. Hope, "Transbilayer Movement and Net Flux of Cholesterol and Cholesterol Sulfate between Liposomal Membranes", Biochemistry (1995) 34, 6208-6217.
30. Sandilands2009
Sandilands A, Sutherland C, Irvine AD, McLean WH, "Filaggrin in the frontline: role in skin barrier function and disease," J Cell Sci. (2009) May 1;122(Pt 9):1285-94.
31. Scappola1995
Scoppola A, Testa G, Frontoni S, Maddaloni E, Gambardella S, Menzinger G and Lala A. "Effects of insulin on cholesterol synthesis in type II diabetes patients," Diabetes Care (1995) 18: pp. 1362-1369.
32. Schallreut94
Schallreuter KU, Wood JM, Pittelkow MR, Gutlich M, Lemke KR, Rodl W, Swanson NN, Hitzemann K, Ziegler I, "Regulation of melanin biosynthesis in the human epidermis by tetrahydrobiopterin." Science (1994) 263(5152);1444-6. PMID: 8128228
33. Seneff2010
S. Seneff, G. Wainwright, and L. Mascitelli, "Is the metabolic syndrome caused by a high fructose, and relatively low fat, low cholesterol diet?", Archives of Medical Science (2010), To Appear.
34. Strott2003
Charles A. Strott and Yuko Higashi, "Cholesterol sulfate in human physiology: what's it all about?" Journal of Lipid Research (2003) Volume 44, pp. 1268-1278.
35. Wahlund1991
Wahlund, T. M., C. R. Woese, R. W. Castenholz, and M. T. Madigan, "A thermophilic green sulfur bacterium from New Zealand hot springs, Chlorobium tepidum sp." Nov. Arch. Microbiol. (1991) 159:81-90.
36. Waldman2009
M. Waldman, MD,, 9th International Conference on Alzheimer's and Parkinson's Diseases (2009) Abstract 90, Presented March 12-13.
37. Wilson2003
Robert F Wilson, Jeffrey F Barletta and James G Tyburski,"Hypocholesterolemia in Sepsis and Critically Ill or Injured Patients" Critical Care 7:413-414, 2003. http://www.medscape.com/viewarticle/511735_2
38. Zerkle2009
Aubrey L. Zerkle, James Farquhar, David T. Johnston, Raymond P. Cox, and Donald E. Canfield, "Fractionation of multiple sulfur isotopes during phototrophic oxidation of sulfide and elemental sulfur by a green sulfur bacterium," Geochimica et Cosmochimica Acta (2009) Volume 73, Issue 2, 15 January 2009, Pages 291-306; doi:10.1016/j.gca.2008.10.027
Thursday, February 25, 2010
Statins and Myoglobin: How Muscle Pain and Weakness Progress to Heart, Lung and Kidney Failure
Statin drug use has steadily increased over the last several decades, due to the widespread belief that cholesterol reduction is an important step in preventing heart disease. It is undeniable that statins are effective: they can decrease serum cholesterol levels from over 300 db/ml to well within the normal range in a matter of weeks. For a person who already has normal cholesterol levels, statins can drive their cholesterol down to levels not seen in nature. Statins have also been shown to reduce the relative risk of heart attacks in men in their 50's by as much as 30%, but, because heart attacks are relatively rare for this segment of the population, the absolute risk reduction is only on the order of 2%, a point that is often missed by the person being treated.
All drugs have potential side effects, and with any drug it's a matter of weighing the risk/benefit factors to decide whether the drug is warranted. Statin drugs have a remarkably diverse set of side effects, including cognitive and memory impairment, reduced libido, and muscle pain and weakness. The drug manufacturers claim that the incidence of side effects is relatively rare, but often side effects don't appear until after several months or even years into treatment. In many of these cases, it may not be obvious that the statin drug is the cause of the problem. This is especially true because these side effects can easily be attributed to increasing age. In fact, as I will show later, statin side effects can best be interpreted as an acceleration of the aging process.
In my view, statin drugs are never worth the risk of their side effects. Cholesterol is a vital nutrient, without which mammalian cells can not survive, and it is inconceivable to me that crippling the body's ability to synthesize cholesterol can ever be a good idea. In an excellent and highly informative review article published in 2009, Wainwright et al. [43] developed a strong argument that statin drugs, by depleting cholesterol, lead to a destabilization of cell membranes "from head to toe." This problem, in turn, increases risk to a long list of serious health conditions and diseases, including diabetes, multiple sclerosis, cognitive problems, hemorrhagic stroke, cancer, and even ALS (amyotrophic lateral sclerosis, also known commonly as Lou Gehrig's disease). Their arguments are backed up by references to 85 peer-reviewed journal publications. I have argued in previous essays that statins may increase the risk to Alzheimer's disease, as well as to sepsis, cancer, and heart failure.
The most commonly reported side effects to statin therapy are muscle pain and weakness. If left unchecked, these symptoms can progress to rhabdomyolysis (severe muscle damage) and kidney failure. Muscle weakness in the lungs can lead to breathing difficulties; in the heart it leads to heart failure. Statin users are reassured by their doctors that they can halt statin therapy if their liver and muscle enzymes rise too high. In practice, however, it's possible to suffer irreversible muscle damage (the problem does not go away after the statin therapy is stopped), and this can happen even when the enzyme levels are not above the normal range.
This essay will develop an argument for why, over time, a statin user may become increasingly weak, in some cases to the point of major disability. A key message is that muscles are forced to cannibalize themselves to acquire sufficient energy. But another factor is oxidative damage to muscle tissue, with subsequent disintegration of the cell walls. This is true not only for the skeletal muscles, but also for the respiratory muscles controlling breathing and the heart muscle. With continued abuse, the muscle cells disintegrate, and the debris travels in the blood stream to the kidneys, which can lead to kidney failure.
The rest of this essay will unfold as follows. In the next section, I will explain how statin drugs work, which will also show why they interfere with the synthesis not only of cholesterol but also of other essential biological substances involved in cell metabolism. The following section will present evidence that statins damage muscle cells. Sections 4 and 5 describe the biochemical pathways involved in assuring that muscles have enough energy to effect movement, especially during situations of stress such as extreme exercise. Section 6 describes the condition known as rhabdomyolysis, caused by extreme exercise but also by statin drugs, and the subsequent risk to kidney failure. Section 7 describes the role that myoglobin, a key protein found in muscle cells, plays in the disease process. After a section explaining how cholesterol protects cell membranes from oxidative damage, the four subsequent sections (sections 9-12) will be devoted to the repercussions of statin damage to the muscles, the heart, the lungs, and the pancreas, respectively. Finally, the conclusion section will summarize the essay and provide hints about my upcoming essay on ALS, a physically disabling neurodegenerative disease that is due not to muscle damage per se but to damage of the motor neurons in the spinal cord that transmit signals from the brain to the skeletal muscles.
All drugs have potential side effects, and with any drug it's a matter of weighing the risk/benefit factors to decide whether the drug is warranted. Statin drugs have a remarkably diverse set of side effects, including cognitive and memory impairment, reduced libido, and muscle pain and weakness. The drug manufacturers claim that the incidence of side effects is relatively rare, but often side effects don't appear until after several months or even years into treatment. In many of these cases, it may not be obvious that the statin drug is the cause of the problem. This is especially true because these side effects can easily be attributed to increasing age. In fact, as I will show later, statin side effects can best be interpreted as an acceleration of the aging process.
In my view, statin drugs are never worth the risk of their side effects. Cholesterol is a vital nutrient, without which mammalian cells can not survive, and it is inconceivable to me that crippling the body's ability to synthesize cholesterol can ever be a good idea. In an excellent and highly informative review article published in 2009, Wainwright et al. [43] developed a strong argument that statin drugs, by depleting cholesterol, lead to a destabilization of cell membranes "from head to toe." This problem, in turn, increases risk to a long list of serious health conditions and diseases, including diabetes, multiple sclerosis, cognitive problems, hemorrhagic stroke, cancer, and even ALS (amyotrophic lateral sclerosis, also known commonly as Lou Gehrig's disease). Their arguments are backed up by references to 85 peer-reviewed journal publications. I have argued in previous essays that statins may increase the risk to Alzheimer's disease, as well as to sepsis, cancer, and heart failure.
The most commonly reported side effects to statin therapy are muscle pain and weakness. If left unchecked, these symptoms can progress to rhabdomyolysis (severe muscle damage) and kidney failure. Muscle weakness in the lungs can lead to breathing difficulties; in the heart it leads to heart failure. Statin users are reassured by their doctors that they can halt statin therapy if their liver and muscle enzymes rise too high. In practice, however, it's possible to suffer irreversible muscle damage (the problem does not go away after the statin therapy is stopped), and this can happen even when the enzyme levels are not above the normal range.
This essay will develop an argument for why, over time, a statin user may become increasingly weak, in some cases to the point of major disability. A key message is that muscles are forced to cannibalize themselves to acquire sufficient energy. But another factor is oxidative damage to muscle tissue, with subsequent disintegration of the cell walls. This is true not only for the skeletal muscles, but also for the respiratory muscles controlling breathing and the heart muscle. With continued abuse, the muscle cells disintegrate, and the debris travels in the blood stream to the kidneys, which can lead to kidney failure.
The rest of this essay will unfold as follows. In the next section, I will explain how statin drugs work, which will also show why they interfere with the synthesis not only of cholesterol but also of other essential biological substances involved in cell metabolism. The following section will present evidence that statins damage muscle cells. Sections 4 and 5 describe the biochemical pathways involved in assuring that muscles have enough energy to effect movement, especially during situations of stress such as extreme exercise. Section 6 describes the condition known as rhabdomyolysis, caused by extreme exercise but also by statin drugs, and the subsequent risk to kidney failure. Section 7 describes the role that myoglobin, a key protein found in muscle cells, plays in the disease process. After a section explaining how cholesterol protects cell membranes from oxidative damage, the four subsequent sections (sections 9-12) will be devoted to the repercussions of statin damage to the muscles, the heart, the lungs, and the pancreas, respectively. Finally, the conclusion section will summarize the essay and provide hints about my upcoming essay on ALS, a physically disabling neurodegenerative disease that is due not to muscle damage per se but to damage of the motor neurons in the spinal cord that transmit signals from the brain to the skeletal muscles.
2. The Biological Mechanism of Statin Drugs
Why do statins cause so many side effects? To answer this question requires explaining all the crucial roles that cholesterol plays in maintaining the integrity and functioning of the body's cells. However, statins interfere not only with the synthesis of cholesterol, but also with the synthesis of an enzyme, coenzyme Q10, that plays a critical role in energy metabolism in all cells. A deficiency in both cholesterol and coenzyme Q10, over time, leads to a huge list of potential health problems. Exactly how an individual responds depends upon their genetic make-up: faced with a deficiency, the body will decide to sacrifice certain cell types in order to safeguard certain other cell types. So, one person may develop Alzheimer's because the brain's neurons are sacrificed, while another succumbs to heart failure or rhabdomyolysis (skeletal muscle wasting).
Statins suppress a critical early step in the multi-step biological pathway that leads to cholesterol synthesis. This is why statins are able to dramatically reduce the blood serum levels of cholesterol. Specifically, statins interfere with the production of the enzyme, HMG-Coenzyme A Reductase, which catalyzes production of mevalonate from its precursor, HMG-Coenzyme A.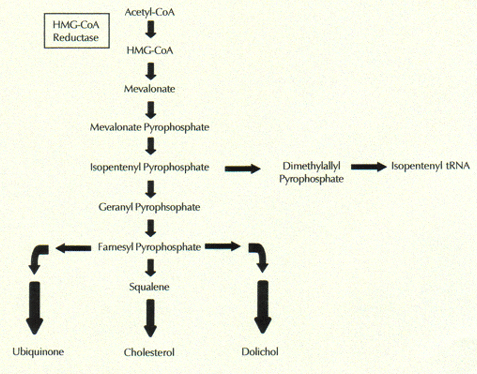 Several more steps produce cholesterol from mevalonate. Mevalonate is also the precursor to a large number of other biologically active molecules that are important for proper cell function. These include the antioxidants, coenzyme Q10 and the dolichols, as shown in the figure to the right.
Several more steps produce cholesterol from mevalonate. Mevalonate is also the precursor to a large number of other biologically active molecules that are important for proper cell function. These include the antioxidants, coenzyme Q10 and the dolichols, as shown in the figure to the right.
The so-called "bad" cholesterol, LDL, delivers cholesterol, fats, and antioxidants from the liver to all the cells of the body. All cells need both fats and cholesterol to maintain healthy membranes, not only in the outer cell wall, but also in the membranes encasing the nucleus, the mitochondria (energy producing units), and the lysosomes (the cell's digestive system). Antioxidants are critical for neutralizing the damaging effects of oxygen exposure, always an issue whenever energy is generated in the mitochondria through a chemical reaction between food sources and oxygen.
In a double-blind placebo controlled study [18], it has been shown that statins can reduce serum levels of coenzyme Q10 by as much as 40%. Coenzyme Q10 is not only a powerful antioxidant, but it also plays a crucial role in the process that breaks down glucose in the presence of oxygen to yield carbon dioxide and water. This metabolic pathway, which is essentially the burning of glucose as fuel, takes place in the mitochondria via the well-known citric acid, or Kreb's cycle. The energy that is released through this process is packaged up in the form of ATP (Adenosine Triphosphate), the currency that all cells use to store their energy reserves.
The dolichols play a special role for the lysosomes [20]. The lysosomes are walled off "rooms" that contain digestive enzymes to break down debris from damaged cell parts so that they can be recycled into useful materials. Lysosomes must maintain a highly acidic internal environment in order for the digestive enzymes to work properly. The dolichols are responsible for pumping hydrogen ions into the lysosomes to keep them highly acidic.
A final way that statins can damage cells is through their entry mechanism. Statins belong to a class of drugs called "amphiphilic" drugs [2] that manage to break through the cell wall in spite of being relatively large. They act like a soap by essentially dissolving a section of the cell membrane. This leaves behind a hole in the wall, which needs to be patched up, as well as debris which must be cleaned up and recycled by the lysosomes. To patch the hole requires new sources of both fats and cholesterol, which come from the LDL particles, whose supply is greatly reduced due to the statin drug. So it becomes increasingly difficult over time for the cell to repair all the holes introduced by the statin drug molecules. As the cell wall becomes more permeable due to previous exposure to an amphiphilic drug, the amount of drug that successfully enters the cell steadily increases over time, leading to ever greater internal concentrations of the drug.
Statins suppress a critical early step in the multi-step biological pathway that leads to cholesterol synthesis. This is why statins are able to dramatically reduce the blood serum levels of cholesterol. Specifically, statins interfere with the production of the enzyme, HMG-Coenzyme A Reductase, which catalyzes production of mevalonate from its precursor, HMG-Coenzyme A.
 Several more steps produce cholesterol from mevalonate. Mevalonate is also the precursor to a large number of other biologically active molecules that are important for proper cell function. These include the antioxidants, coenzyme Q10 and the dolichols, as shown in the figure to the right.
Several more steps produce cholesterol from mevalonate. Mevalonate is also the precursor to a large number of other biologically active molecules that are important for proper cell function. These include the antioxidants, coenzyme Q10 and the dolichols, as shown in the figure to the right. The so-called "bad" cholesterol, LDL, delivers cholesterol, fats, and antioxidants from the liver to all the cells of the body. All cells need both fats and cholesterol to maintain healthy membranes, not only in the outer cell wall, but also in the membranes encasing the nucleus, the mitochondria (energy producing units), and the lysosomes (the cell's digestive system). Antioxidants are critical for neutralizing the damaging effects of oxygen exposure, always an issue whenever energy is generated in the mitochondria through a chemical reaction between food sources and oxygen.
In a double-blind placebo controlled study [18], it has been shown that statins can reduce serum levels of coenzyme Q10 by as much as 40%. Coenzyme Q10 is not only a powerful antioxidant, but it also plays a crucial role in the process that breaks down glucose in the presence of oxygen to yield carbon dioxide and water. This metabolic pathway, which is essentially the burning of glucose as fuel, takes place in the mitochondria via the well-known citric acid, or Kreb's cycle. The energy that is released through this process is packaged up in the form of ATP (Adenosine Triphosphate), the currency that all cells use to store their energy reserves.
The dolichols play a special role for the lysosomes [20]. The lysosomes are walled off "rooms" that contain digestive enzymes to break down debris from damaged cell parts so that they can be recycled into useful materials. Lysosomes must maintain a highly acidic internal environment in order for the digestive enzymes to work properly. The dolichols are responsible for pumping hydrogen ions into the lysosomes to keep them highly acidic.
A final way that statins can damage cells is through their entry mechanism. Statins belong to a class of drugs called "amphiphilic" drugs [2] that manage to break through the cell wall in spite of being relatively large. They act like a soap by essentially dissolving a section of the cell membrane. This leaves behind a hole in the wall, which needs to be patched up, as well as debris which must be cleaned up and recycled by the lysosomes. To patch the hole requires new sources of both fats and cholesterol, which come from the LDL particles, whose supply is greatly reduced due to the statin drug. So it becomes increasingly difficult over time for the cell to repair all the holes introduced by the statin drug molecules. As the cell wall becomes more permeable due to previous exposure to an amphiphilic drug, the amount of drug that successfully enters the cell steadily increases over time, leading to ever greater internal concentrations of the drug.
3. Statins, Muscle Pain and Weakness, and Rhabdomyolysis
Muscle cells have tremendous energy needs, particularly if the person has been put on an exercise regimen as part of their treatment program. The heart, in particular, never rests. It has to keep on beating 24x7 at the rate of at least once every second. Hence the heart is especially dependent on coenzyme Q10 to replenish the ATP consumed every time it contracts and pushes blood from one chamber to another and out the aorta.
The pharmaceutical industry readily admits that statin therapy may cause muscle pain and/or muscle weakness in some cases, but they claim that the incidence of these side effects is small, on the order of 2%. However, observational studies have shown that at least 10% to 15% of statin users complain of muscle pain [6][40]. The actual number who experience pain or weakness is likely to be much larger, however, because many people are unaware that this is a potential side effect. Furthermore, it sometimes takes several years of cumulative statin damage before the symptoms become intolerable. People are often willing to believe that their aches and pains and generally weakening condition are simply due to getting older.
The reaction by the general community to a relatively benign article posted by WebMD on muscle pain suggests that the problem is much worse than is generally acknowledged. Over 200 often lengthy comments describe many very sad stories; often the doctor was also misinformed, and denied that the pain could be due to the statin drug. A case in point is described in this New York Times article. A woman in Kansas had been taking a statin for years to reduce her cholesterol. Over that same time period, she experienced chronic muscle pain which neither she nor her doctor attributed to the statin therapy. It even led to a useless shoulder operation. Her problem eventually escalated into skin lesions caused by a reaction to toxic protein by-products released by her disintegrating muscles. She was given an antifungal to treat the skin lesions, another misdiagnosis. But the antifungal interacted with the statins [25] to further increase the severity of her muscle disorders. Three months later, she could barely stand, and her pulmonary muscles were so weak she couldn't breathe. She died shortly thereafter.
Rhabdomyolysis is a condition where the muscles rapidly disintegrate due to an injury, often, for example, physical trauma following an accident. But Rhabdomyolysis is also a rare side effect of statins -- essentially where the muscle pain and weakness are extreme. Some people react immediately to statin therapy with severe rhabdomyolysis, and it is often fatal, due to acute renal failure (ARF). Myoglobin is sloughed off from the muscle cells in large amounts, and it overloads the kidneys and causes them to shut down completely. Initiating statin therapy is therefore a bit like Russian roulette -- there is even a known case where a single statin dose caused rhabdomyolysis [21]. One of the statins, Baycol, was abruptly taken off the market in 2001, after 31 people died from subsequent rhabdomyolysis.
The pharmaceutical industry readily admits that statin therapy may cause muscle pain and/or muscle weakness in some cases, but they claim that the incidence of these side effects is small, on the order of 2%. However, observational studies have shown that at least 10% to 15% of statin users complain of muscle pain [6][40]. The actual number who experience pain or weakness is likely to be much larger, however, because many people are unaware that this is a potential side effect. Furthermore, it sometimes takes several years of cumulative statin damage before the symptoms become intolerable. People are often willing to believe that their aches and pains and generally weakening condition are simply due to getting older.
The reaction by the general community to a relatively benign article posted by WebMD on muscle pain suggests that the problem is much worse than is generally acknowledged. Over 200 often lengthy comments describe many very sad stories; often the doctor was also misinformed, and denied that the pain could be due to the statin drug. A case in point is described in this New York Times article. A woman in Kansas had been taking a statin for years to reduce her cholesterol. Over that same time period, she experienced chronic muscle pain which neither she nor her doctor attributed to the statin therapy. It even led to a useless shoulder operation. Her problem eventually escalated into skin lesions caused by a reaction to toxic protein by-products released by her disintegrating muscles. She was given an antifungal to treat the skin lesions, another misdiagnosis. But the antifungal interacted with the statins [25] to further increase the severity of her muscle disorders. Three months later, she could barely stand, and her pulmonary muscles were so weak she couldn't breathe. She died shortly thereafter.
Rhabdomyolysis is a condition where the muscles rapidly disintegrate due to an injury, often, for example, physical trauma following an accident. But Rhabdomyolysis is also a rare side effect of statins -- essentially where the muscle pain and weakness are extreme. Some people react immediately to statin therapy with severe rhabdomyolysis, and it is often fatal, due to acute renal failure (ARF). Myoglobin is sloughed off from the muscle cells in large amounts, and it overloads the kidneys and causes them to shut down completely. Initiating statin therapy is therefore a bit like Russian roulette -- there is even a known case where a single statin dose caused rhabdomyolysis [21]. One of the statins, Baycol, was abruptly taken off the market in 2001, after 31 people died from subsequent rhabdomyolysis.
4. How Muscles Maintain their Energy Supply
In this section and the next, I will describe the metabolic pathways involved in assuring that muscle cells have enough energy to contract. When oxygen is available, and when it can be safely exploited, the muscle can decompose food sources into carbon dioxide and water, by consuming the oxygen. But oxygen, while life-giving, is also a very dangerous substance, and if the process is not orchestrated exactly according to plan, there can be a lot of collateral damage due to the wrong substances reacting with the oxygen. As you will see later, myoglobin, which is responsible for buffering oxygen and delivering it from the cell wall to the mitochondria, picks up a lot of the collateral damage. This aerobic metabolic process is termed respiration, and it takes place inside special organelles called mitochondria, whose sole responsibility is to digest foods and generate energy for the cell.
Whenever oxygen is in short supply, or if the mitochondria are dysfunctional, the cell has alternative ways to generate energy (e.g., fermentation), which take place, in the absence of oxygen, in the main compartment of the cell, called the cytoplasm. These processes require exchanges of nutrients between the muscles and the liver, and they require the assistance of special enzymes which then show up in the blood stream. These are the very same enzymes whose concentrations are monitored to detect whether a statin drug may have damaged the muscles.
If you don't feel compelled to know the details of how all these processes work, you could skip this section and section 5, and, I think, still be able to follow the rest of the story.
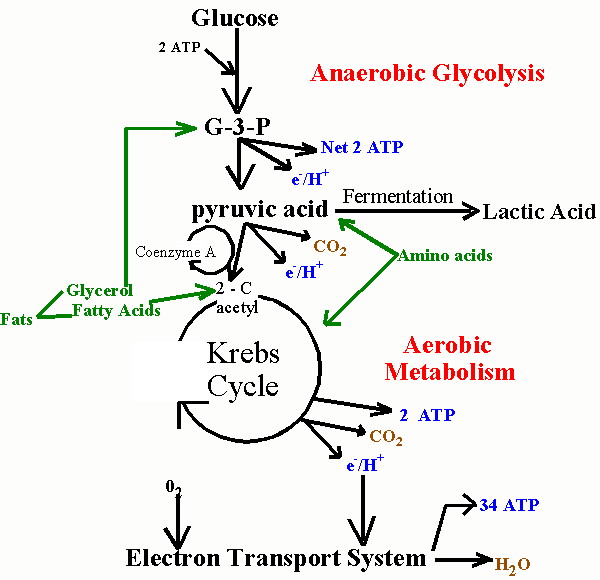 In order to explain how statins damage muscles, I will first need to explain how muscles manage their energy needs. Muscles require a significant amount of energy to contract, and they get most of this energy by breaking down fatty acids and glucose obtained originally from food sources. Like all eukaryotic cells (cells containing a nucleus), muscle cells are able to generate a lot of energy through aerobic (oxygen-requiring) processes that are sequestered within special energy-generating subregions of the cell called mitochondria. This aerobic metabolic process is highly efficient, generating as many as 30 units of ATP (Adenosine Triphosphate) for each molecule of glucose. ATP can be thought of as an energy currency, because it can be easily broken down to AMP (adenosine monophosphate), releasing the stored energy in the process, which will then fuel cell contraction.
In order to explain how statins damage muscles, I will first need to explain how muscles manage their energy needs. Muscles require a significant amount of energy to contract, and they get most of this energy by breaking down fatty acids and glucose obtained originally from food sources. Like all eukaryotic cells (cells containing a nucleus), muscle cells are able to generate a lot of energy through aerobic (oxygen-requiring) processes that are sequestered within special energy-generating subregions of the cell called mitochondria. This aerobic metabolic process is highly efficient, generating as many as 30 units of ATP (Adenosine Triphosphate) for each molecule of glucose. ATP can be thought of as an energy currency, because it can be easily broken down to AMP (adenosine monophosphate), releasing the stored energy in the process, which will then fuel cell contraction.
Unfortunately, the process of metabolizing food sources for energy is quite complex. I have found two images which depict food metabolism in complementary ways, where one (above, right) shows chemical reactions and the other (below, left) schematizes the regions of the cell that are involved. They use slightly different nomenclature, but I will try to link them together when necessary. When glucose first enters the cell (mediated through insulin), it is converted to pyruvate (also called pyruvic acid) in the cell's cytoplasm (the main compartment of the cell). This process releases a small amount of ATP, but does not require oxygen, which makes it useful when oxygen is in short supply. The pyruvate can also be broken down to lactate (also called lactic acid) (fermentation, oxygen absent in the figure below) in the cytoplasm, without requiring any oxygen, so-called anaerobic metabolism, to release additional energy. This pathway is important to muscle cells under conditions of extreme exercise, when oxygen supplies become depleted.
To generate a much larger amount of ATP requires the help of the mitochondria (the large oval purple-shaped object in the figure), and involves a well known process referred to multiple ways: as the respiratory or electron transport chain, the Tricarboxylic acid (TCA) cycle or the Krebs Cycle (TCA cycle, oxygen present in the figure below; Krebs Cycle, Aerobic Metabolism in the figure above). The process is tricky, because oxygen molecules (O2) have to be split apart, and, during intermediate stages, dangerous free radicals are lying around (individual negatively charged oxygen atoms that have not yet fully combined with hydrogen (H+) to form the very stable molecule, water (H2O)).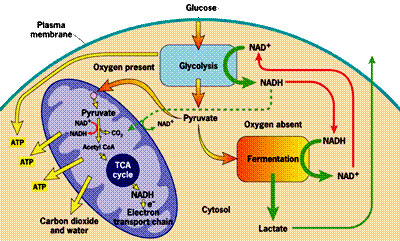 These free radicals are highly reactive. Antioxidants are compounds that can absorb these free radicals and render them harmless. Two very important antioxidants that play a critical role in the electron transport chain are coenzyme Q10 (also known as ubiquinone) and cytochrome c.
These free radicals are highly reactive. Antioxidants are compounds that can absorb these free radicals and render them harmless. Two very important antioxidants that play a critical role in the electron transport chain are coenzyme Q10 (also known as ubiquinone) and cytochrome c.
 The small figure to the left shows a schematic of a mitochondrion, and the larger figure below shows a more detailed explanation of the electron transport chain process that takes place along the wall enclosing the mitochondrion, generating a large percentage of the cell's energy needs in the process. The electron transport chain injects protons (H+) into the intermembrane space, essentially creating a battery (charge differential across the membrane) that can then complete the process of converting (spent) AMP back to ATP as a renewed energy source. If there is an insufficient supply of coenzyme Q10 (also known as "ubiquinone", the "Q" in the figure), then the
The small figure to the left shows a schematic of a mitochondrion, and the larger figure below shows a more detailed explanation of the electron transport chain process that takes place along the wall enclosing the mitochondrion, generating a large percentage of the cell's energy needs in the process. The electron transport chain injects protons (H+) into the intermembrane space, essentially creating a battery (charge differential across the membrane) that can then complete the process of converting (spent) AMP back to ATP as a renewed energy source. If there is an insufficient supply of coenzyme Q10 (also known as "ubiquinone", the "Q" in the figure), then the 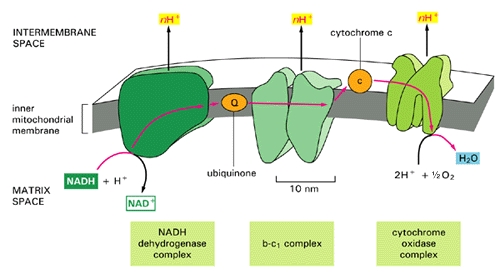 electron transport chain will not work as efficiently. Hydrogen ions will leak back into the mitochondrion through a passive process, requiring a much larger expenditure of energy to push them back out [20]. The battery charge will be reduced, and there will be a decrease in the amount of ATP that can be generated. The net effect will be very similar to the effect of insufficient oxygen, with respect to energy generated. However, it will be much more damaging because, instead of being absent, the oxygen is present but is only partially converted to water (2H+ + 1/2 O2 -> H2O at the right of the figure), since the chain of events is held up at the "Q" position. Various highly toxic charged ions containing oxygen, such as -OH, H2O2 (hydrogen peroxide) and *OH, will linger and wreak havoc on the muscle cell, as you will see later.
electron transport chain will not work as efficiently. Hydrogen ions will leak back into the mitochondrion through a passive process, requiring a much larger expenditure of energy to push them back out [20]. The battery charge will be reduced, and there will be a decrease in the amount of ATP that can be generated. The net effect will be very similar to the effect of insufficient oxygen, with respect to energy generated. However, it will be much more damaging because, instead of being absent, the oxygen is present but is only partially converted to water (2H+ + 1/2 O2 -> H2O at the right of the figure), since the chain of events is held up at the "Q" position. Various highly toxic charged ions containing oxygen, such as -OH, H2O2 (hydrogen peroxide) and *OH, will linger and wreak havoc on the muscle cell, as you will see later.
There are a number of rare genetic disorders that involve mutations in genes encoding enzymes that operate within the electron transport chain [23][32]. Particularly relevant to our story are Complex I enzymes, because Coenzyme Q10 is one of them. An interesting case study involved two sisters [23], both of whom suffered from a genetic mutation leading to a defect identified to be associated with the NADH-Coenzyme Q10 complex. As would be predicted, they suffered from substantially decreased rates of respiratory metabolism (the process discussed above). They also were extremely weak and had marked intolerance to exercise. When they exercised, their levels of lactate and pyruvate rose sharply in the blood, an indication that they were relying on anaerobic fermentation in the cytoplasm rather than aerobic metabolism in the mitochondria to meet their energy needs.
Whenever oxygen is in short supply, or if the mitochondria are dysfunctional, the cell has alternative ways to generate energy (e.g., fermentation), which take place, in the absence of oxygen, in the main compartment of the cell, called the cytoplasm. These processes require exchanges of nutrients between the muscles and the liver, and they require the assistance of special enzymes which then show up in the blood stream. These are the very same enzymes whose concentrations are monitored to detect whether a statin drug may have damaged the muscles.
If you don't feel compelled to know the details of how all these processes work, you could skip this section and section 5, and, I think, still be able to follow the rest of the story.
 In order to explain how statins damage muscles, I will first need to explain how muscles manage their energy needs. Muscles require a significant amount of energy to contract, and they get most of this energy by breaking down fatty acids and glucose obtained originally from food sources. Like all eukaryotic cells (cells containing a nucleus), muscle cells are able to generate a lot of energy through aerobic (oxygen-requiring) processes that are sequestered within special energy-generating subregions of the cell called mitochondria. This aerobic metabolic process is highly efficient, generating as many as 30 units of ATP (Adenosine Triphosphate) for each molecule of glucose. ATP can be thought of as an energy currency, because it can be easily broken down to AMP (adenosine monophosphate), releasing the stored energy in the process, which will then fuel cell contraction.
In order to explain how statins damage muscles, I will first need to explain how muscles manage their energy needs. Muscles require a significant amount of energy to contract, and they get most of this energy by breaking down fatty acids and glucose obtained originally from food sources. Like all eukaryotic cells (cells containing a nucleus), muscle cells are able to generate a lot of energy through aerobic (oxygen-requiring) processes that are sequestered within special energy-generating subregions of the cell called mitochondria. This aerobic metabolic process is highly efficient, generating as many as 30 units of ATP (Adenosine Triphosphate) for each molecule of glucose. ATP can be thought of as an energy currency, because it can be easily broken down to AMP (adenosine monophosphate), releasing the stored energy in the process, which will then fuel cell contraction.Unfortunately, the process of metabolizing food sources for energy is quite complex. I have found two images which depict food metabolism in complementary ways, where one (above, right) shows chemical reactions and the other (below, left) schematizes the regions of the cell that are involved. They use slightly different nomenclature, but I will try to link them together when necessary. When glucose first enters the cell (mediated through insulin), it is converted to pyruvate (also called pyruvic acid) in the cell's cytoplasm (the main compartment of the cell). This process releases a small amount of ATP, but does not require oxygen, which makes it useful when oxygen is in short supply. The pyruvate can also be broken down to lactate (also called lactic acid) (fermentation, oxygen absent in the figure below) in the cytoplasm, without requiring any oxygen, so-called anaerobic metabolism, to release additional energy. This pathway is important to muscle cells under conditions of extreme exercise, when oxygen supplies become depleted.
To generate a much larger amount of ATP requires the help of the mitochondria (the large oval purple-shaped object in the figure), and involves a well known process referred to multiple ways: as the respiratory or electron transport chain, the Tricarboxylic acid (TCA) cycle or the Krebs Cycle (TCA cycle, oxygen present in the figure below; Krebs Cycle, Aerobic Metabolism in the figure above). The process is tricky, because oxygen molecules (O2) have to be split apart, and, during intermediate stages, dangerous free radicals are lying around (individual negatively charged oxygen atoms that have not yet fully combined with hydrogen (H+) to form the very stable molecule, water (H2O)).
 These free radicals are highly reactive. Antioxidants are compounds that can absorb these free radicals and render them harmless. Two very important antioxidants that play a critical role in the electron transport chain are coenzyme Q10 (also known as ubiquinone) and cytochrome c.
These free radicals are highly reactive. Antioxidants are compounds that can absorb these free radicals and render them harmless. Two very important antioxidants that play a critical role in the electron transport chain are coenzyme Q10 (also known as ubiquinone) and cytochrome c.  The small figure to the left shows a schematic of a mitochondrion, and the larger figure below shows a more detailed explanation of the electron transport chain process that takes place along the wall enclosing the mitochondrion, generating a large percentage of the cell's energy needs in the process. The electron transport chain injects protons (H+) into the intermembrane space, essentially creating a battery (charge differential across the membrane) that can then complete the process of converting (spent) AMP back to ATP as a renewed energy source. If there is an insufficient supply of coenzyme Q10 (also known as "ubiquinone", the "Q" in the figure), then the
The small figure to the left shows a schematic of a mitochondrion, and the larger figure below shows a more detailed explanation of the electron transport chain process that takes place along the wall enclosing the mitochondrion, generating a large percentage of the cell's energy needs in the process. The electron transport chain injects protons (H+) into the intermembrane space, essentially creating a battery (charge differential across the membrane) that can then complete the process of converting (spent) AMP back to ATP as a renewed energy source. If there is an insufficient supply of coenzyme Q10 (also known as "ubiquinone", the "Q" in the figure), then the  electron transport chain will not work as efficiently. Hydrogen ions will leak back into the mitochondrion through a passive process, requiring a much larger expenditure of energy to push them back out [20]. The battery charge will be reduced, and there will be a decrease in the amount of ATP that can be generated. The net effect will be very similar to the effect of insufficient oxygen, with respect to energy generated. However, it will be much more damaging because, instead of being absent, the oxygen is present but is only partially converted to water (2H+ + 1/2 O2 -> H2O at the right of the figure), since the chain of events is held up at the "Q" position. Various highly toxic charged ions containing oxygen, such as -OH, H2O2 (hydrogen peroxide) and *OH, will linger and wreak havoc on the muscle cell, as you will see later.
electron transport chain will not work as efficiently. Hydrogen ions will leak back into the mitochondrion through a passive process, requiring a much larger expenditure of energy to push them back out [20]. The battery charge will be reduced, and there will be a decrease in the amount of ATP that can be generated. The net effect will be very similar to the effect of insufficient oxygen, with respect to energy generated. However, it will be much more damaging because, instead of being absent, the oxygen is present but is only partially converted to water (2H+ + 1/2 O2 -> H2O at the right of the figure), since the chain of events is held up at the "Q" position. Various highly toxic charged ions containing oxygen, such as -OH, H2O2 (hydrogen peroxide) and *OH, will linger and wreak havoc on the muscle cell, as you will see later. There are a number of rare genetic disorders that involve mutations in genes encoding enzymes that operate within the electron transport chain [23][32]. Particularly relevant to our story are Complex I enzymes, because Coenzyme Q10 is one of them. An interesting case study involved two sisters [23], both of whom suffered from a genetic mutation leading to a defect identified to be associated with the NADH-Coenzyme Q10 complex. As would be predicted, they suffered from substantially decreased rates of respiratory metabolism (the process discussed above). They also were extremely weak and had marked intolerance to exercise. When they exercised, their levels of lactate and pyruvate rose sharply in the blood, an indication that they were relying on anaerobic fermentation in the cytoplasm rather than aerobic metabolism in the mitochondria to meet their energy needs.
5. Managing Energy Needs During Extreme Exercise
Whenever a healthy person experiences extreme exercise, such as running a 500 meter dash, the muscles are challenged to come up with sufficient ATP to satisfy their energy needs. Both oxygen and glucose can become depleted. To compensate for these deficiencies, muscle cells have devised a complex set of strategies, which operate in the cytoplasm instead of in the mitochondria. They involve a number of enzymes that will resurface later in our story, since they are the enzymes that are monitored to determine whether statin drugs are damaging muscles and/or the liver.
As you've already seen, one option is to generate lactic acid anaerobically (without consuming oxygen), but this still provides only 1/6 as much ATP as the aerobic process taking place in the mitochondria. The process of generating energy from ATP happens in two steps: ATP is first converted to ADP (adenosine diphosphate) and finally to AMP (adenosine monophosphate). When excess amounts of AMP accumulate in the muscle cell, the cell is induced to take up extra glucose, which will soon deplete the supply (of glucose) in the blood unless the liver can efficiently regenerate more. ADP can be converted back to ATP with the help of an enzyme, creatine kinase. In addition, the conversion of pyruvate (generated anaerobically from glucose) to lactate requires the help of another enzyme, lactate dehydrogenase. Lactate builds up when oxygen is insufficient, and is released into the blood stream. Fortunately, the heart is able to utilize lactate as an alternative fuel source [9], which becomes especially important during times of extreme exercise.
In order for the liver to generate more glucose, it needs a substrate. In the short term, muscles can supply this substrate, but it requires self cannibalization. During brief periods of starvation, human muscle cells adapt quickly by breaking down muscle proteins and converting them into a basic amino acid, alanine [34]. Muscles then rely on a novel mechanism that involves an exchange system with the liver, the so-called glucose-alanine cycle. The alanine, derived from muscle protein, is released into the blood and shipped to the liver to be utilized for energy generation, as shown in the accompanying figure. The liver can then generate more glucose from the alanine through gluconeogenesis, while exporting the waste product, urea, to the kidneys for excretion. This also allows the liver to regenerate some ATP to help satisfy its own energy needs, which are very large during such stressful conditions. The glucose is shipped through the blood stream to the muscle cell, which eagerly takes it up to generate more ATP for itself. The anaerobic processing of glucose yields pyruvate which can also be turned into alanine, but it needs yet another enzyme to work. Thus, whenever pyruvate can't be sent to the mitochondria because of insufficient oxygen, it can instead be converted to alanine with the help of an enzyme, ALT (alanine aminotransferase), as long as there is a good supply of glutamate, which is converted to alpha-keto glutarate in the process.
as long as there is a good supply of glutamate, which is converted to alpha-keto glutarate in the process.
In the above discussion, several enzymes have been identified that must be present for these cytoplasmic energy-generating processes to function. These include creatine kinase, lactate dehydrogenase, and ALT. The so-called liver enzyme test that is conducted routinely with statin users measures the concentration of ALT in the blood. Muscle enzyme tests detect creatine kinase and lactate dehydrogenase concentrations in the blood. So these tests are all measuring these particular enzymes because they signal that muscles are preferentially processing glucose anaerobically in the cytoplasm instead of aerobically in the mitochondria; i.e., the mitochondria are not working properly. You should keep this point in mind as we will revisit it later on.
As you've already seen, one option is to generate lactic acid anaerobically (without consuming oxygen), but this still provides only 1/6 as much ATP as the aerobic process taking place in the mitochondria. The process of generating energy from ATP happens in two steps: ATP is first converted to ADP (adenosine diphosphate) and finally to AMP (adenosine monophosphate). When excess amounts of AMP accumulate in the muscle cell, the cell is induced to take up extra glucose, which will soon deplete the supply (of glucose) in the blood unless the liver can efficiently regenerate more. ADP can be converted back to ATP with the help of an enzyme, creatine kinase. In addition, the conversion of pyruvate (generated anaerobically from glucose) to lactate requires the help of another enzyme, lactate dehydrogenase. Lactate builds up when oxygen is insufficient, and is released into the blood stream. Fortunately, the heart is able to utilize lactate as an alternative fuel source [9], which becomes especially important during times of extreme exercise.
In order for the liver to generate more glucose, it needs a substrate. In the short term, muscles can supply this substrate, but it requires self cannibalization. During brief periods of starvation, human muscle cells adapt quickly by breaking down muscle proteins and converting them into a basic amino acid, alanine [34]. Muscles then rely on a novel mechanism that involves an exchange system with the liver, the so-called glucose-alanine cycle. The alanine, derived from muscle protein, is released into the blood and shipped to the liver to be utilized for energy generation, as shown in the accompanying figure. The liver can then generate more glucose from the alanine through gluconeogenesis, while exporting the waste product, urea, to the kidneys for excretion. This also allows the liver to regenerate some ATP to help satisfy its own energy needs, which are very large during such stressful conditions. The glucose is shipped through the blood stream to the muscle cell, which eagerly takes it up to generate more ATP for itself. The anaerobic processing of glucose yields pyruvate which can also be turned into alanine, but it needs yet another enzyme to work. Thus, whenever pyruvate can't be sent to the mitochondria because of insufficient oxygen, it can instead be converted to alanine with the help of an enzyme, ALT (alanine aminotransferase),
 as long as there is a good supply of glutamate, which is converted to alpha-keto glutarate in the process.
as long as there is a good supply of glutamate, which is converted to alpha-keto glutarate in the process. In the above discussion, several enzymes have been identified that must be present for these cytoplasmic energy-generating processes to function. These include creatine kinase, lactate dehydrogenase, and ALT. The so-called liver enzyme test that is conducted routinely with statin users measures the concentration of ALT in the blood. Muscle enzyme tests detect creatine kinase and lactate dehydrogenase concentrations in the blood. So these tests are all measuring these particular enzymes because they signal that muscles are preferentially processing glucose anaerobically in the cytoplasm instead of aerobically in the mitochondria; i.e., the mitochondria are not working properly. You should keep this point in mind as we will revisit it later on.
6. Extreme Exercise can lead to Rhabdomyolysis
When people engage in extreme exercise such as long distance marathon running or weight-bearing exercises, they run the risk of causing serious damage, both to their muscles and to their kidneys, due to the stress imposed on their system in trying to maintain adequate energy to fuel the muscles. It has become common practice to measure the levels of creatine kinase in the blood as a known indicator of potential damage [5]. A person whose creatine kinase level gets alarmingly high will likely need immediate medical attention to avoid renal [kidney] failure.
The cause of the kidney failure is most likely myoglobin that has been dumped into the blood stream by compromised or dead muscle cells, due to rhabdomyolysis. If too much myoglobin is released, particularly with inadequate water supply, the myoglobin can block the renal filtration system causing a condition known as "acute tubular necrosis." The problem can be easily detected by observing the color of the urine, which will be dark brown. A study published in 2009 showed that, in rhabdomyolysis, the damage to the kidneys involves direct interaction between myoglobin and the mitochondria in the kidney cells [33]. The resulting oxidation of the mitochondrial membranes leads to respiratory failure and subsequent cell death.
Myoglobinuria is the term used to describe the presence of myoglobin in the urine, usually due to rhabdomyolysis. According to [37], 15% of patients with severe myoglobinuria develop acute renal failure, and it is associated with high mortality rates. Dialysis or intravenous fluids must be introduced fast enough, or the person will not be able to recover.
As early as 1991, a group of Japanese researchers [38] demonstrated that coenzyme Q10 could be administered orally to protect rats from muscle damage due to strenuous exercise. They also noticed that rats that were administered coenzyme Q10 did not have elevated levels of creatine kinase and lactate dehydrogenase, whereas the control rats did.
The cause of the kidney failure is most likely myoglobin that has been dumped into the blood stream by compromised or dead muscle cells, due to rhabdomyolysis. If too much myoglobin is released, particularly with inadequate water supply, the myoglobin can block the renal filtration system causing a condition known as "acute tubular necrosis." The problem can be easily detected by observing the color of the urine, which will be dark brown. A study published in 2009 showed that, in rhabdomyolysis, the damage to the kidneys involves direct interaction between myoglobin and the mitochondria in the kidney cells [33]. The resulting oxidation of the mitochondrial membranes leads to respiratory failure and subsequent cell death.
Myoglobinuria is the term used to describe the presence of myoglobin in the urine, usually due to rhabdomyolysis. According to [37], 15% of patients with severe myoglobinuria develop acute renal failure, and it is associated with high mortality rates. Dialysis or intravenous fluids must be introduced fast enough, or the person will not be able to recover.
As early as 1991, a group of Japanese researchers [38] demonstrated that coenzyme Q10 could be administered orally to protect rats from muscle damage due to strenuous exercise. They also noticed that rats that were administered coenzyme Q10 did not have elevated levels of creatine kinase and lactate dehydrogenase, whereas the control rats did.
7. Myoglobin: The Good, The Bad, and the Ugly
Myoglobin is a unique protein specially adapted for muscle cells to help them with their enormous needs for oxygen. Its physical structure is schematized in the figure on the right. It resembles hemoglobin in that it contains a central heme element (schematized in red in the figure) whose active  ingredient is a single charged iron atom (Fe). While hemoglobin, found in red blood cells, transports oxygen from the lungs to all the tissues of the body, myoglobin functions to store excess oxygen within the muscle cell, to help buffer supply during periods of excessive demand. It also transports oxygen from the cell wall to the mitochondria. Even with help from the myoglobin, it's often the case that muscles have to resort to anaerobic metabolism under strenuous exercise, during which lactic acid is built up and released into the blood stream.
ingredient is a single charged iron atom (Fe). While hemoglobin, found in red blood cells, transports oxygen from the lungs to all the tissues of the body, myoglobin functions to store excess oxygen within the muscle cell, to help buffer supply during periods of excessive demand. It also transports oxygen from the cell wall to the mitochondria. Even with help from the myoglobin, it's often the case that muscles have to resort to anaerobic metabolism under strenuous exercise, during which lactic acid is built up and released into the blood stream.
Myoglobin exists in at least three distinct forms, which can be characterized as Mg+2 (Ferrous), Mg+3 (Ferric), and Mg+4 (Ferryl), depending upon the amount of charge that is present on the central Iron atom. As Mg+2, its healthy state, it will readily take up oxygen and store it, whereas, when converted to Mg+3 by the addition of a proton, it becomes inert. However, with the addition of yet another proton, it becomes Mg+4, a highly toxic reactive agent that will begin to break down the fatty acids contained in the outer cell wall of the muscle cell (so-called peroxidative damage)[35], and go on to destroy the cholesterol in the cell wall as well [31]. Myoglobin becomes Ferryl myoglobin in the presence of excess amounts of free radicals, i.e., under oxidative stress induced by highly reactive oxygen compounds like hydrogen peroxide. Recall that, with statin therapy, hydrogen peroxide is generated in the mitochondria because the process of breaking down oxygen and converting it to water is incomplete -- due to the insufficient supply of coenzyme Q10.
An excellent article describing the process by which a cell is injured by oxidative stress was written by John Farber in 1994 [13]. He wrote: "All aerobic cells generate, enzymatically or nonenzymatically, a constitutive flux of O2-, H2O2, and possibly *OH. At the same time, the abundant antioxidant defenses of most cells, again both enzymatic and nonenzymatic, prevent these species from causing cell injury. Nevertheless, there are situations in which the rate of formation of partially reduced oxygen species is increased and/or the antioxidant defenses of the cells are weakened. In either case, oxidative cell injury may result." [14, p. 17]. The process of aerobic oxidation of food sources to generate energy is confined to the mitochondria in order to protect the constituents in the cytoplasm as much as possible. But myoglobin is tasked with transporting oxygen from the cell wall through the cytoplasm to the mitochondria. It can't avoid oxygen exposure, and when it delivers the oxygen, it necessarily has to come in contact with these toxic intermediate products of the process that ultimately converts oxygen to water. One of the most important roles of coenzyme Q10 in the muscle cells is to neutralize the damage to myoglobin caused by these oxidative agents.
When a person suffers from a heart attack (ischemic event), their muscles experience an extreme lack of oxygen due to the heart's temporary inability to pump blood. However, one of the most dangerous aspects of a heart attack is the so-called reperfusion period, when blood circulation is restored, but after the cells have suffered injury as a consequence of oxygen deprivation [29]. This condition is especially problematic for the heart muscle, since it is so crucial to survival. In a study involving rats that had suffered from heart attacks, it was proposed that the injury is a direct consequence of exposure to the Fe+4 form of myoglobin (ferryl myoglobin) [1]. Because the cells have been unable to maintain their physiological state during the deprivation period, they are highly vulnerable to oxidative stress.
Once the fatty acids in a muscle's cell wall are broken down due to exposure to toxic Ferryl myoglobin, the cell rapidly disintegrates. Because the cell wall is no longer impermeable to ions, large amounts of calcium start rushing into the cell, and soon after it dies [14]. The debris of the dead and dying cells gets dispersed into the blood stream, and makes its way to the kidneys for disposal. This causes a tremendous load on the kidneys which can sometimes lead to their failure as well [47], and the situation cascades into a downward spiral.
In 1994, Mordente et al. published a paper that investigated in vitro the degree to which coenzyme Q could protect myoglobin from oxidative damage [28]. Their results showed convincingly that coenzyme Q can work as a natural antioxidant for myoglobin. To quote the last sentence of their abstract: "Collectively, these studies suggest that the proposed function of coenzyme Q as a naturally occurring antioxidant might well relate to its ability of reducing H2O2 [hydrogen peroxide]-activated myoglobin. Coenzyme Q should therefore mitigate cardiac or muscular dysfunctions that are caused by an abnormal generation of H2O2."
 ingredient is a single charged iron atom (Fe). While hemoglobin, found in red blood cells, transports oxygen from the lungs to all the tissues of the body, myoglobin functions to store excess oxygen within the muscle cell, to help buffer supply during periods of excessive demand. It also transports oxygen from the cell wall to the mitochondria. Even with help from the myoglobin, it's often the case that muscles have to resort to anaerobic metabolism under strenuous exercise, during which lactic acid is built up and released into the blood stream.
ingredient is a single charged iron atom (Fe). While hemoglobin, found in red blood cells, transports oxygen from the lungs to all the tissues of the body, myoglobin functions to store excess oxygen within the muscle cell, to help buffer supply during periods of excessive demand. It also transports oxygen from the cell wall to the mitochondria. Even with help from the myoglobin, it's often the case that muscles have to resort to anaerobic metabolism under strenuous exercise, during which lactic acid is built up and released into the blood stream. Myoglobin exists in at least three distinct forms, which can be characterized as Mg+2 (Ferrous), Mg+3 (Ferric), and Mg+4 (Ferryl), depending upon the amount of charge that is present on the central Iron atom. As Mg+2, its healthy state, it will readily take up oxygen and store it, whereas, when converted to Mg+3 by the addition of a proton, it becomes inert. However, with the addition of yet another proton, it becomes Mg+4, a highly toxic reactive agent that will begin to break down the fatty acids contained in the outer cell wall of the muscle cell (so-called peroxidative damage)[35], and go on to destroy the cholesterol in the cell wall as well [31]. Myoglobin becomes Ferryl myoglobin in the presence of excess amounts of free radicals, i.e., under oxidative stress induced by highly reactive oxygen compounds like hydrogen peroxide. Recall that, with statin therapy, hydrogen peroxide is generated in the mitochondria because the process of breaking down oxygen and converting it to water is incomplete -- due to the insufficient supply of coenzyme Q10.
An excellent article describing the process by which a cell is injured by oxidative stress was written by John Farber in 1994 [13]. He wrote: "All aerobic cells generate, enzymatically or nonenzymatically, a constitutive flux of O2-, H2O2, and possibly *OH. At the same time, the abundant antioxidant defenses of most cells, again both enzymatic and nonenzymatic, prevent these species from causing cell injury. Nevertheless, there are situations in which the rate of formation of partially reduced oxygen species is increased and/or the antioxidant defenses of the cells are weakened. In either case, oxidative cell injury may result." [14, p. 17]. The process of aerobic oxidation of food sources to generate energy is confined to the mitochondria in order to protect the constituents in the cytoplasm as much as possible. But myoglobin is tasked with transporting oxygen from the cell wall through the cytoplasm to the mitochondria. It can't avoid oxygen exposure, and when it delivers the oxygen, it necessarily has to come in contact with these toxic intermediate products of the process that ultimately converts oxygen to water. One of the most important roles of coenzyme Q10 in the muscle cells is to neutralize the damage to myoglobin caused by these oxidative agents.
When a person suffers from a heart attack (ischemic event), their muscles experience an extreme lack of oxygen due to the heart's temporary inability to pump blood. However, one of the most dangerous aspects of a heart attack is the so-called reperfusion period, when blood circulation is restored, but after the cells have suffered injury as a consequence of oxygen deprivation [29]. This condition is especially problematic for the heart muscle, since it is so crucial to survival. In a study involving rats that had suffered from heart attacks, it was proposed that the injury is a direct consequence of exposure to the Fe+4 form of myoglobin (ferryl myoglobin) [1]. Because the cells have been unable to maintain their physiological state during the deprivation period, they are highly vulnerable to oxidative stress.
Once the fatty acids in a muscle's cell wall are broken down due to exposure to toxic Ferryl myoglobin, the cell rapidly disintegrates. Because the cell wall is no longer impermeable to ions, large amounts of calcium start rushing into the cell, and soon after it dies [14]. The debris of the dead and dying cells gets dispersed into the blood stream, and makes its way to the kidneys for disposal. This causes a tremendous load on the kidneys which can sometimes lead to their failure as well [47], and the situation cascades into a downward spiral.
In 1994, Mordente et al. published a paper that investigated in vitro the degree to which coenzyme Q could protect myoglobin from oxidative damage [28]. Their results showed convincingly that coenzyme Q can work as a natural antioxidant for myoglobin. To quote the last sentence of their abstract: "Collectively, these studies suggest that the proposed function of coenzyme Q as a naturally occurring antioxidant might well relate to its ability of reducing H2O2 [hydrogen peroxide]-activated myoglobin. Coenzyme Q should therefore mitigate cardiac or muscular dysfunctions that are caused by an abnormal generation of H2O2."
8. How Cholesterol Protects Membranes and Saves Energy
Mammalian cells can not survive without cholesterol [45]. Cholesterol is found in the outer wall (cell membrane) of all cells in the body. It is also found in internal membranes that surround both the mitochondria and the lysosomes (highly acidic containers of digestive 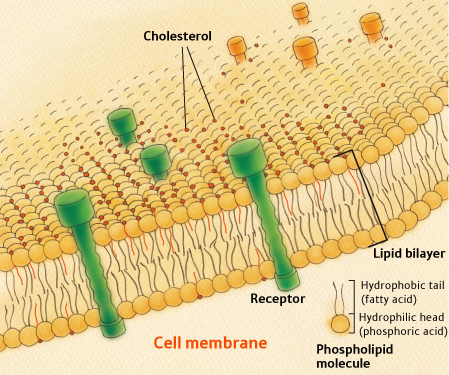 enzymes). To understand how cholesterol works, you need to know something about the structure of cell membranes. All cell membranes are built from a so-called lipid bilayer, as illustrated in the figure to the right. The lipid bilayer contains two parallel chains of phospholipids (the same phospholipids that encase LDL particles, the so-called "bad" cholesterol). Phospholipids have the unique property that one end of the molecule is hydrophobic (water insoluble) and the other is hydrophilic (water soluble). The two chains in the lipid bilayer orient themselves such that the hydrophobic sides of both layers are adjoining in the center of the membrane. This central hydrophobic layer thus contains fatty acids that are vulnerable to oxidative damage. The outer parts, facing both the exterior and the interior of the cell, are water-soluble. Cholesterol molecules are dispersed throughout the membrane at strategic locations.
enzymes). To understand how cholesterol works, you need to know something about the structure of cell membranes. All cell membranes are built from a so-called lipid bilayer, as illustrated in the figure to the right. The lipid bilayer contains two parallel chains of phospholipids (the same phospholipids that encase LDL particles, the so-called "bad" cholesterol). Phospholipids have the unique property that one end of the molecule is hydrophobic (water insoluble) and the other is hydrophilic (water soluble). The two chains in the lipid bilayer orient themselves such that the hydrophobic sides of both layers are adjoining in the center of the membrane. This central hydrophobic layer thus contains fatty acids that are vulnerable to oxidative damage. The outer parts, facing both the exterior and the interior of the cell, are water-soluble. Cholesterol molecules are dispersed throughout the membrane at strategic locations.
An article published in 2009 by Kucerka et al. [22] nicely sums up several known roles of cholesterol in membranes: "Cholesterol is found in all animal cell membranes and is required for proper membrane permeability and fluidity. It is also needed for building and maintaining cell membranes, and may act as an antioxidant. Recently, cholesterol has also been implicated in cell signaling processes, and is suggested to enable lipid raft formation in the plasma membrane." [Ibid, p. 16358] The article goes on to describe how cholesterol is able to orient itself within the membrane either vertically (bridging across the membrane) or horizontally (sequestered within the hydrophobic central space of the membrane lipid bilayer). How it is oriented depends upon the degree to which the fatty acids in the membrane are saturated, with saturated fatty acids greatly favoring the vertical over the horizontal orientation. Cholesterol can also flip easily from one side of the bilayer to the other. All of this flexibility in its orientation within the membrane allows it to operate effectively as a signaling molecule.
A fascinating article written by Thomas Haines in 2001 proposes a novel but compelling role for cholesterol in protecting the cell membrane from sodium leaks [20]. All mammalian cells maintain an ion gradient across their outer wall, which is utilized to fuel cell chemical processes. The so-called sodium pump is an active process that constantly pumps sodium out of the cell to maintain this charge difference. The pump consumes ATP in the process. Working against the pump is a passive leakage mechanism that causes sodium to drift back into the cell. To the extent that the membrane can be constructed to resist leakage (kind of like putting insulation in the attic of a house), it will require less ATP to maintain sodium concentrations appropriate for the cell to function properly.
The Haines article argues that cholesterol plays an essential role in protecting the cell wall from sodium leakage. Sodium leakage is a much bigger problem (it leaks 7 to 11 times as fast in the absence of cholesterol) for unsaturated fatty acids as for saturated fatty acids [4]. However, unsaturated fatty acids also encourage cholesterol to arrange itself in the central layer. By accumulating there, it provides extra insulation preventing the charged sodium ions from passively hopping from the exterior to the interior of the cell. Other experiments [30] have shown that the relative sodium leakage rates are reduced by 300% in the presence of cholesterol.
 enzymes). To understand how cholesterol works, you need to know something about the structure of cell membranes. All cell membranes are built from a so-called lipid bilayer, as illustrated in the figure to the right. The lipid bilayer contains two parallel chains of phospholipids (the same phospholipids that encase LDL particles, the so-called "bad" cholesterol). Phospholipids have the unique property that one end of the molecule is hydrophobic (water insoluble) and the other is hydrophilic (water soluble). The two chains in the lipid bilayer orient themselves such that the hydrophobic sides of both layers are adjoining in the center of the membrane. This central hydrophobic layer thus contains fatty acids that are vulnerable to oxidative damage. The outer parts, facing both the exterior and the interior of the cell, are water-soluble. Cholesterol molecules are dispersed throughout the membrane at strategic locations.
enzymes). To understand how cholesterol works, you need to know something about the structure of cell membranes. All cell membranes are built from a so-called lipid bilayer, as illustrated in the figure to the right. The lipid bilayer contains two parallel chains of phospholipids (the same phospholipids that encase LDL particles, the so-called "bad" cholesterol). Phospholipids have the unique property that one end of the molecule is hydrophobic (water insoluble) and the other is hydrophilic (water soluble). The two chains in the lipid bilayer orient themselves such that the hydrophobic sides of both layers are adjoining in the center of the membrane. This central hydrophobic layer thus contains fatty acids that are vulnerable to oxidative damage. The outer parts, facing both the exterior and the interior of the cell, are water-soluble. Cholesterol molecules are dispersed throughout the membrane at strategic locations.An article published in 2009 by Kucerka et al. [22] nicely sums up several known roles of cholesterol in membranes: "Cholesterol is found in all animal cell membranes and is required for proper membrane permeability and fluidity. It is also needed for building and maintaining cell membranes, and may act as an antioxidant. Recently, cholesterol has also been implicated in cell signaling processes, and is suggested to enable lipid raft formation in the plasma membrane." [Ibid, p. 16358] The article goes on to describe how cholesterol is able to orient itself within the membrane either vertically (bridging across the membrane) or horizontally (sequestered within the hydrophobic central space of the membrane lipid bilayer). How it is oriented depends upon the degree to which the fatty acids in the membrane are saturated, with saturated fatty acids greatly favoring the vertical over the horizontal orientation. Cholesterol can also flip easily from one side of the bilayer to the other. All of this flexibility in its orientation within the membrane allows it to operate effectively as a signaling molecule.
A fascinating article written by Thomas Haines in 2001 proposes a novel but compelling role for cholesterol in protecting the cell membrane from sodium leaks [20]. All mammalian cells maintain an ion gradient across their outer wall, which is utilized to fuel cell chemical processes. The so-called sodium pump is an active process that constantly pumps sodium out of the cell to maintain this charge difference. The pump consumes ATP in the process. Working against the pump is a passive leakage mechanism that causes sodium to drift back into the cell. To the extent that the membrane can be constructed to resist leakage (kind of like putting insulation in the attic of a house), it will require less ATP to maintain sodium concentrations appropriate for the cell to function properly.
The Haines article argues that cholesterol plays an essential role in protecting the cell wall from sodium leakage. Sodium leakage is a much bigger problem (it leaks 7 to 11 times as fast in the absence of cholesterol) for unsaturated fatty acids as for saturated fatty acids [4]. However, unsaturated fatty acids also encourage cholesterol to arrange itself in the central layer. By accumulating there, it provides extra insulation preventing the charged sodium ions from passively hopping from the exterior to the interior of the cell. Other experiments [30] have shown that the relative sodium leakage rates are reduced by 300% in the presence of cholesterol.
Subscribe to:
Posts (Atom)